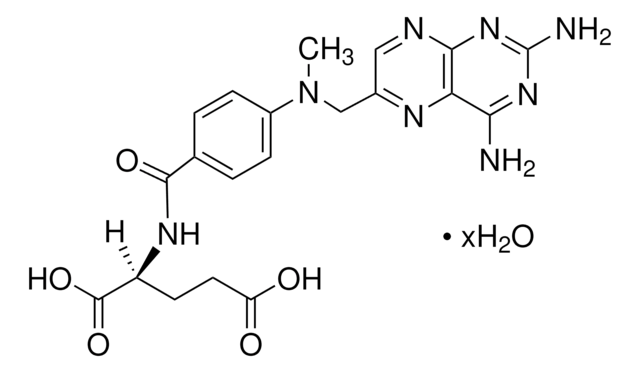
G2536
Ganciclovir
≥99% (HPLC), powder, viral DNA elongation inhibitor
Synonym(s):
2-Amino-1,9-dihydro-9-[[2-hydroxy-1-(hydroxymethyl)ethoxy]methyl]-6H-purin-6-one, 9-(1,3-Dihydroxy-2-propoxymethyl)guanine, DHPG
About This Item
Recommended Products
Product Name
Ganciclovir, ≥99% (HPLC), powder
Quality Level
assay
≥99% (HPLC)
form
powder
color
white
solubility
0.1 M HCl: 10 mg/mL
ε (extinction coefficient)
12.0 at 256 nm at 1 mM
antibiotic activity spectrum
viruses
mode of action
DNA synthesis | interferes
originator
Roche
storage temp.
2-8°C
SMILES string
NC1=Nc2c(ncn2COC(CO)CO)C(=O)N1
InChI
1S/C9H13N5O4/c10-9-12-7-6(8(17)13-9)11-3-14(7)4-18-5(1-15)2-16/h3,5,15-16H,1-2,4H2,(H3,10,12,13,17)
InChI key
IRSCQMHQWWYFCW-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
Biochem/physiol Actions
Upon expression of a viral suicide gene encoding thymidine kinase, the non-toxic pro-drug is converted to a phosphorylated active analog and is incorporated into the DNA of replicating eukaryotic cells, causing death of the malignant dividing cell. The cell cycle is irreversibly arrested at the G2-M checkpoint. Gap junction involvement in the ganciclovir bystander effect has been studied. Ganciclovir has been used to study loss of telomeres and to evaluate sensitivity of viruses to antiviral treatments.
Features and Benefits
Preparation Note
This product should be stored desiccated at 2-8 °C. Under these conditions the product is stable for 3 years.
signalword
Danger
hcodes
Hazard Classifications
Muta. 1B - Repr. 2
Storage Class
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type P3 (EN 143) respirator cartridges
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
MISSION® Target ID Library for Human miRNA Target Identification and Discovery;
DNA damage and repair mechanism is vital for maintaining DNA integrity. Damage to cellular DNA is involved in mutagenesis, the development of cancer among others.
Related Content
We offer agonists, antagonists, modulators and other bioactive small molecules for immune system signaling target identification and validation, as well as a variety of antibiotics, antivirals, and antifungals.
Discover Bioactive Small Molecules for ADME/Tox
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service