F1147
Fumonisin B1 from Fusarium moniliforme
≥98% (HPLC)
Synonym(s):
Macrofusine
About This Item
Recommended Products
biological source
Fusarium sp. (Fusarium moniliforme)
Quality Level
assay
≥98% (HPLC)
form
powder
solubility
methanol: 9.80-10.20 mg/mL, clear, colorless to light yellow
storage temp.
2-8°C
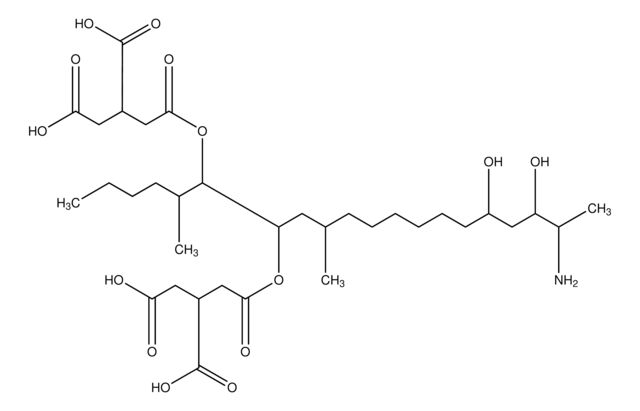
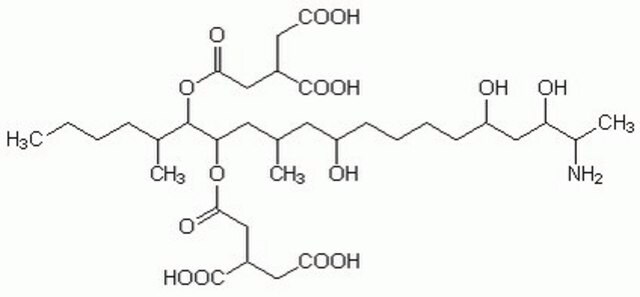
SMILES string
CCCC[C@@H](C)[C@@H](OC(=O)C[C@@H](CC(O)=O)C(O)=O)[C@H](C[C@@H](C)C[C@H](O)CCCC[C@@H](O)C[C@H](O)[C@H](C)N)OC(=O)C[C@@H](CC(O)=O)C(O)=O
InChI
1S/C34H59NO15/c1-5-6-9-20(3)32(50-31(44)17-23(34(47)48)15-29(41)42)27(49-30(43)16-22(33(45)46)14-28(39)40)13-19(2)12-24(36)10-7-8-11-25(37)18-26(38)21(4)35/h19-27,32,36-38H,5-18,35H2,1-4H3,(H,39,40)(H,41,42)(H,45,46)(H,47,48)/t19-,20+,21-,22+,23+,24+,25+,26-,27-,32+/m0/s1
InChI key
UVBUBMSSQKOIBE-DSLOAKGESA-N
Looking for similar products? Visit Product Comparison Guide
General description
Fumonisin B1 (FB1) shares structure similarities with sphingosine. Thus fumonisin B1 can inhibit ceramide synthase (sphingosine N-acetyltransferase), which can lead to disruption of ceramide biosynthesis and complex sphingolipid biosynthesis.
Application
- as a standard in the analysis of mycotoxins in caecal content obtained from adult pigs
- in the study of the effect of fumonisin B1 on the metabolism of prion protein (PrP) isoforms and the synthesis of scrapie prion protein PrPSc and
- in the study of the toxic effects of fumonisin B1 on explants obtained from pig jejunum.
Biochem/physiol Actions
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 3 Oral - Carc. 2 - Repr. 2 - STOT RE 2
target_organs
Kidney,Liver
Storage Class
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type P3 (EN 143) respirator cartridges
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service