E5036
Epidermal Growth Factor Protein, human
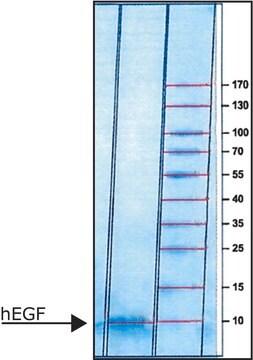
>97% (SDS-PAGE), recombinant, expressed in E. coli, lyophilized powder, suitable for cell culture
Synonym(s):
Epidermal Growth Factor human, EGF
About This Item
Recommended Products
product name
Epidermal Growth Factor, human, animal component free, EGF, recombinant, expressed in Escherichia coli, >97% (SDS-PAGE)
biological source
human
Quality Level
recombinant
expressed in E. coli
assay
>97% (SDS-PAGE)
form
lyophilized powder
potency
0.08-0.8 ng/mL EC50
mol wt
~6 kDa
packaging
pkg of 200 and 500 μg
storage condition
avoid repeated freeze/thaw cycles
impurities
≤1 EU/μg Endotoxin
color
white
solubility
water: soluble 0.190-0.210, clear, colorless
UniProt accession no.
storage temp.
−20°C
Gene Information
human ... EGF(1950)
Looking for similar products? Visit Product Comparison Guide
General description
Application
- as a supplement in in LHC-8 medium to culture liver cell lines
- in the fetal bovine serum (FBS)-Dulbecco′s modified essential medium (DMEM) /F12 medium for primary culture of human glioma cells
- as an additive in the conditional medium of normal fibroblasts (NFs) to study its effect on the migration and invasion of endometrial cancer (EC) cells
- as a component in tumorsphere medium
Biochem/physiol Actions
Citation
2. Gregory, H., Isolation and structure of urogastrone and its relationship to epidermal growth factor. Nature, 257, 325-327 (1975).
3. George-Nascimento, C. et al., Characterization of recombinant human epidermal growth factor produced in yeast. Biochemistry, 27, 797-802 (1988).
4. Todaro, G.J. et al., Transforming growth factors produced by certain human tumor cells: polypeptides that interact with epidermal growth factor receptors. Proc. Natl. Acad. Sci. USA, 77, 5258-5262 (1980).
5. Blomquist, M.C. et al., Vaccinia virus 19-kilodalton protein: relationship to several mammalian proteins, including two growth factors. Proc. Natl. Acad. Sci. USA, 81, 7363-7367 (1984).
6. Eppstein, D.A. et al., Epidermal growth factor receptor occupancy inhibits vaccinia virus infection. Nature, 318, 663-665 (1985).
Storage Class
13 - Non Combustible Solids
wgk_germany
WGK 2
flash_point_f
Not applicable
flash_point_c
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service