C-12375
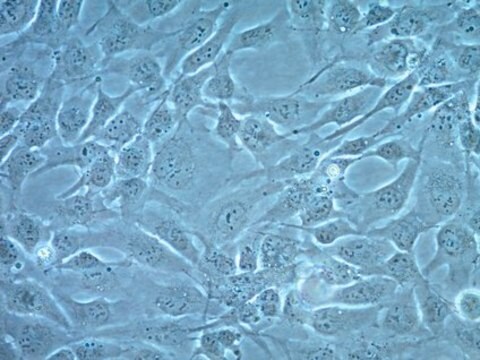
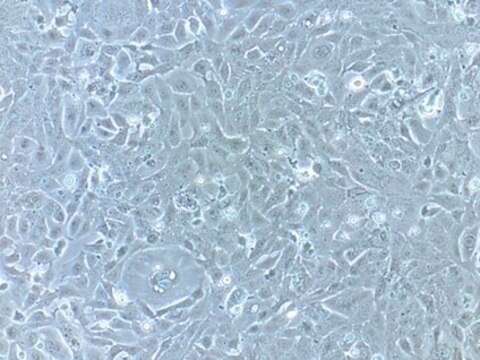
Human Cardiac Fibroblasts (HCF)
500,000 cryopreserved cells
Synonym(s):
HCF cells
About This Item
Recommended Products
biological source
human heart (ventricles)
packaging
pkg of 500,000 cells
morphology
(fibroblast)
technique(s)
cell culture | mammalian: suitable
relevant disease(s)
cardiovascular diseases
shipped in
dry ice
storage temp.
−196°C
General description
Cell Line Origin
Application
Quality
Warning
Subculture Routine
Other Notes
Recommended products
Disclaimer
Storage Class
12 - Non Combustible Liquids
wgk_germany
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
Choose from one of the most recent versions:
Certificates of Analysis (COA)
It looks like we've run into a problem, but you can still download Certificates of Analysis from our Documents section.
If you need assistance, please contact Customer Support.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Articles
Primary human fibroblasts and growth medium for the in vitro cultivation of juvenile and adult fibroblasts. Protocols for handling, cell passaging, media, and product use.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service