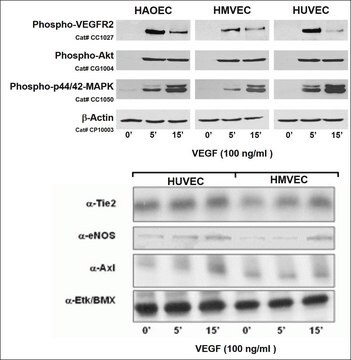
C-12203
Human Umbilical Vein Endothelial Cells (HUVEC)
Pooled, 500,000 cryopreserved cells
Synonym(s):
HUVEC cells
About This Item
Recommended Products
biological source
human umbilical cord vein
packaging
pkg of 500,000 cells
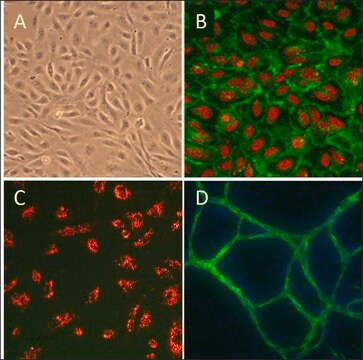
morphology
( endothelial)
technique(s)
cell culture | mammalian: suitable
shipped in
dry ice
storage temp.
−196°C
General description
Cell Line Origin
Application
Quality
Warning
Subculture Routine
Other Notes
Recommended products
Disclaimer
Storage Class
12 - Non Combustible Liquids
wgk_germany
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Sorry, we don't have COAs for this product available online at this time.
If you need assistance, please contact Customer Support.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Protocols
Cell culture protocol: the endothelial cell transwell migration and invasion assay used to study angiogenesis and cancer cell metastasis. Explore over 350 PromoCell products.
Related Content
Cell culture protocol: the endothelial tube formation assay to study angiogenesis using HUVECs and other endothelial cell types. Explore over 350 PromoCell products.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service