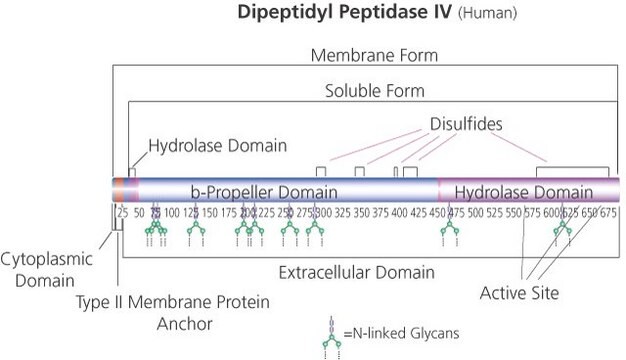
A6778
Angiotensin Converting Enzyme from rabbit lung
≥2.0 units/mg protein (modified Warburg-Christian)
Synonym(s):
ACE, Peptidyl-dipeptidase A, Peptidyldipeptide Hydrolase
About This Item
Recommended Products
form
lyophilized powder
Quality Level
specific activity
≥2.0 units/mg protein (modified Warburg-Christian)
shipped in
dry ice
storage temp.
−20°C
Looking for similar products? Visit Product Comparison Guide
General description
Application
- for measuring inhibitory effect of egg white protein hydrolysates on ACE activity by high performance liquid chromatography (HPLC)
- to measure the ACE inhibition by litchi pericarp and cooked chicken breast using hippuryl-L-histidyl-L-leucine (HHL) as substrate by reverse phase-HPLC (RP-HPLC)3 and HPLC respectively
- in releasing GPI anchored protein in vitro in few cell lines like HeLa, HEK293 and in vivo in mice sperm.
Biochem/physiol Actions
Quality
Unit Definition
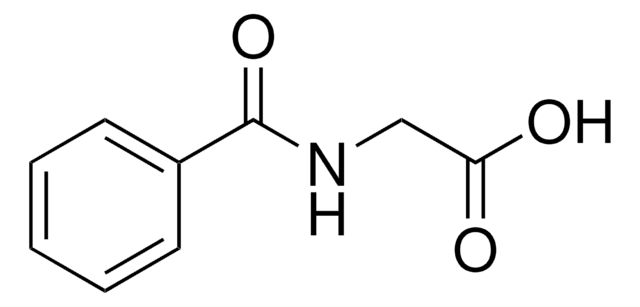
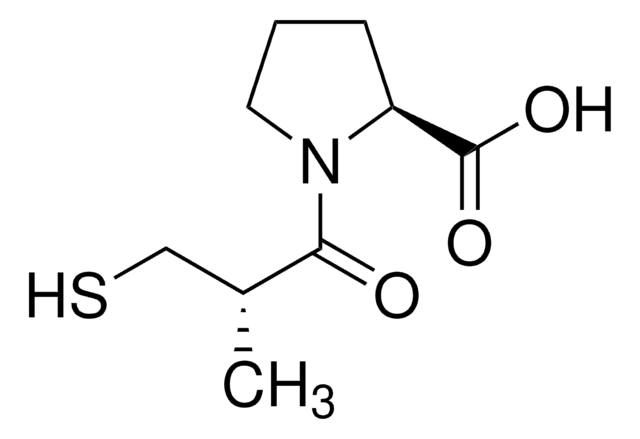
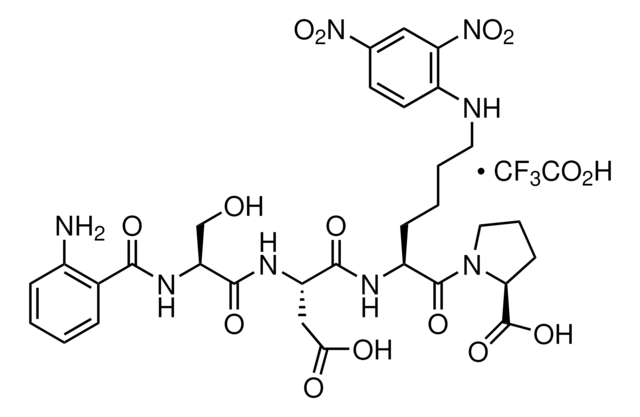
inhibitor
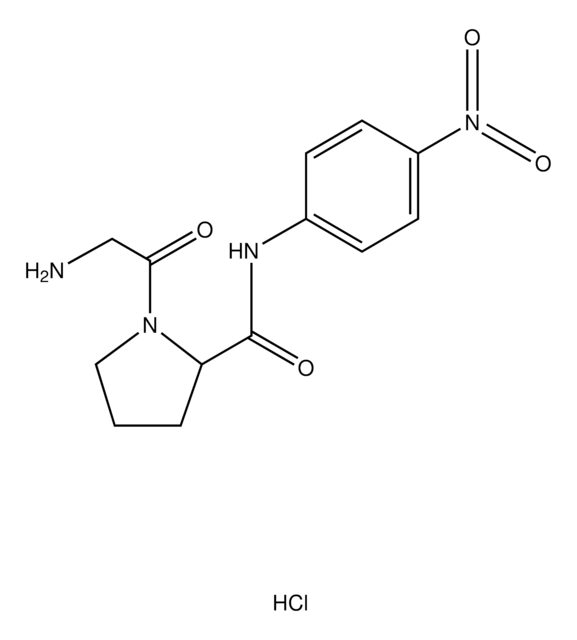
substrate
signalword
Danger
hcodes
pcodes
Hazard Classifications
Resp. Sens. 1
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service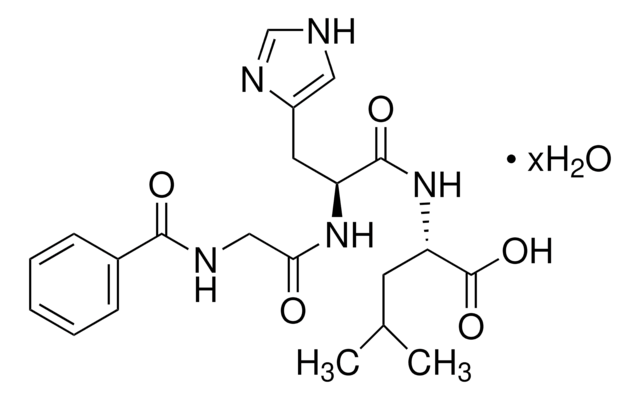




![N-[3-(2-Furyl)acryloyl]-Phe-Gly-Gly](/deepweb/assets/sigmaaldrich/product/structures/225/349/530bc714-b1a8-4fdb-8082-a39329ee730a/640/530bc714-b1a8-4fdb-8082-a39329ee730a.png)