90081
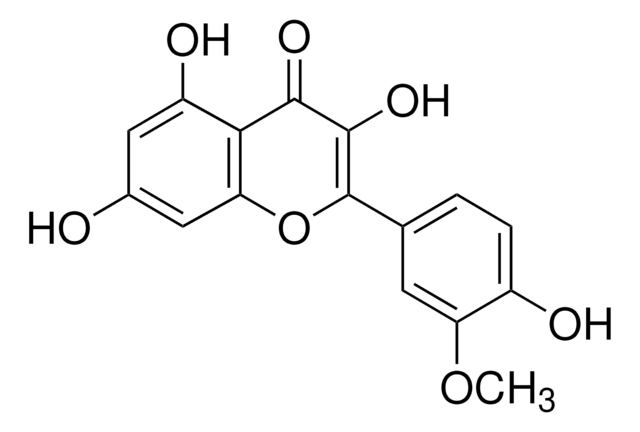
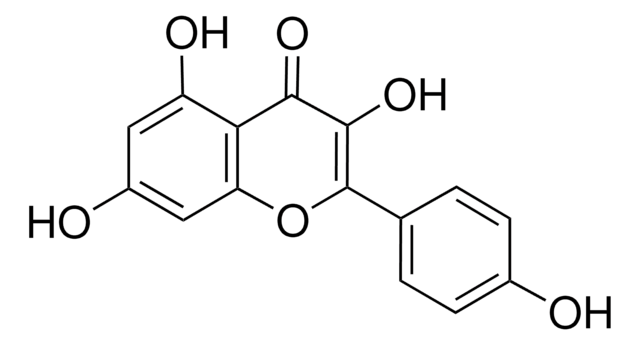
3-O-Methylquercetin
≥97% (HPLC)
Synonym(s):
5,7,3′ ,4′ -Tetrahydroxy-3-methoxyflavone, Quercetin 3-O-methyl ether
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C16H12O7
CAS Number:
Molecular Weight:
316.26
Beilstein/REAXYS Number:
324509
MDL number:
UNSPSC Code:
12352202
PubChem Substance ID:
NACRES:
NA.32
Recommended Products
Quality Level
assay
≥97% (HPLC)
form
powder
SMILES string
COC1=C(Oc2cc(O)cc(O)c2C1=O)c3ccc(O)c(O)c3
InChI
1S/C16H12O7/c1-22-16-14(21)13-11(20)5-8(17)6-12(13)23-15(16)7-2-3-9(18)10(19)4-7/h2-6,17-20H,1H3
InChI key
WEPBGSIAWZTEJR-UHFFFAOYSA-N
Biochem/physiol Actions
3-O-Methylquercetin significantly inhibits cyclic adenosine monophosphate- (cAMP-) and cyclic guanosine monophosphate- (cGMP-) phosphodiesterase activity. It possess anti-inflammatory, bronchodilating properties and used in treatment of asthma. It suppresses the total inflammatory cells, tumor necrosis factor-α (TNF-α) and attenuates the production of interleukins.
3-O-Methylquercetin is a metabolite in flavone and flavonol biosynthesis. It is a naturally occurring compound present in various plants, and was shown to have potent anticancer-promoting, antioxidant, antiallergy, and antimicrobial activity, and showed strong anti-viral activity inhibition of tomato ringspot virus.
Packaging
Bottomless glass bottle. Contents are inside inserted fused cone.
signalword
Danger
hcodes
pcodes
Hazard Classifications
Acute Tox. 3 Oral
Storage Class
6.1C - Combustible, acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
3-O-methylquercetin more selectively inhibits phosphodiesterase subtype 3
Ko WC, et al.
Planta Medica, 69(04), 310-315 (2003)
Quercetin-3-methyl ether suppresses proliferation of mouse epidermal JB6 P+ cells by targeting ERKs.
Jixia Li et al.
Carcinogenesis, 33(2), 459-465 (2011-12-06)
Chemoprevention has been acknowledged as an important and practical strategy for the management of skin cancer. Quercetin-3-methyl ether, a naturally occurring compound present in various plants, has potent anticancer-promoting activity. We identified this compound by in silico virtual screening of
Ana Paula Preczenhak et al.
Food chemistry, 286, 600-607 (2019-03-05)
This study investigated the effectiveness of cysteine in conservation of bioactive compounds and the antioxidant capacity of minimally processed red beet. After red beet minimal processing increasing cysteine concentrations were applied, corresponding to control, 2 mM, 4 mM, 8 mM and 16 mM. Assay
C Angeloni et al.
Biochimie, 89(1), 73-82 (2006-10-19)
The aim of this study was to investigate the potential of quercetin and two of its "in vivo" metabolites, 3'-O-methyl quercetin and 4'-O-methyl quercetin, to protect H9c2 cardiomyoblasts against H(2)O(2)-induced oxidative stress. As limited data are available regarding the potential
Clement K Ameho et al.
The Journal of nutritional biochemistry, 19(7), 467-474 (2007-10-02)
Dietary antioxidants interact in a dynamic fashion, including recycling and sparing one another, to decrease oxidative stress. Limited information is available regarding the interrelationships in vivo between quercetin and vitamin E. We investigated the antioxidant activity and metabolism of quercetin
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service