49101
β-Glucanase from Aspergillus niger
powder, dark brown, ~1 U/mg
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Recommended Products
form
powder
Quality Level
specific activity
~1 U/mg
greener alternative product characteristics
Waste Prevention
Design for Energy Efficiency
Learn more about the Principles of Green Chemistry.
sustainability
Greener Alternative Product
color
dark brown
greener alternative category
storage temp.
2-8°C
Related Categories
General description
We are committed to bringing you Greener Alternative Products, which adhere to one or more of The 12 Principles of Greener Chemistry. This product has been enhanced for energy eficiency and waste prevention when used in cellulosic ethanol research. For more information see the article in biofiles.
Biochem/physiol Actions
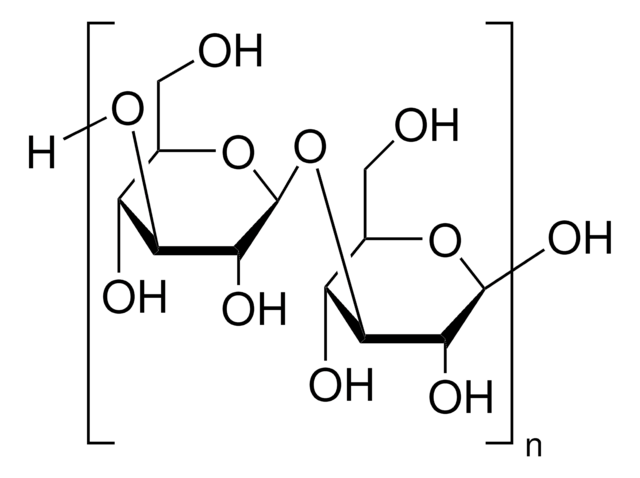
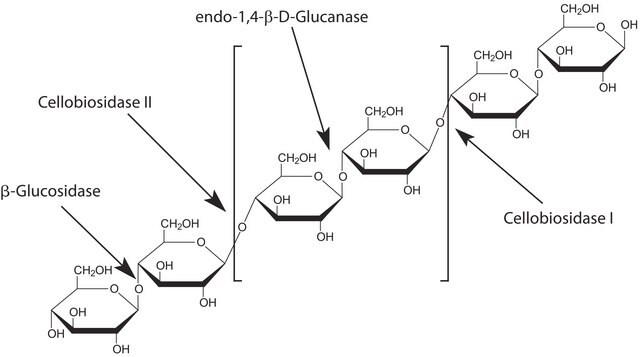
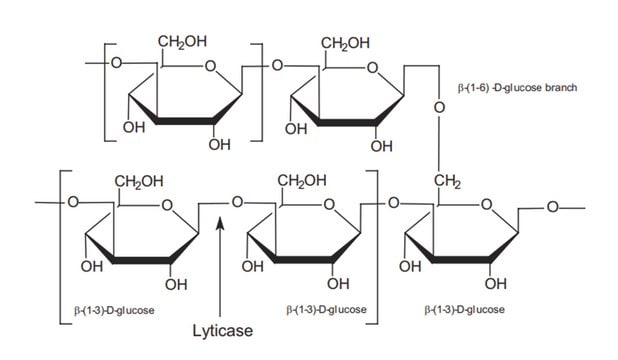
β-glucanases degrade β-1,4-glucans of cellulose, xyloglucan and β-1,4-xylan.
Unit Definition
One unit corresponds to the amount of enzyme which will release 1 μmole of reducing sugar equivalents (expressed as glucose) per minute at pH 5.0 and 55 °C, using β-D-glucan (Cat. No. 49102) as substrate
signalword
Danger
hcodes
pcodes
Hazard Classifications
Resp. Sens. 1
Storage Class
11 - Combustible Solids
wgk_germany
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Yigang Teng et al.
Acta biochimica et biophysica Sinica, 42(10), 729-734 (2010-09-28)
In this study, we confirmed that at least three endo-β-1,4-glucanases existed in the digestive juice of the giant snail, Achatina fulica ferussac, by Congo red staining assay. One of these enzymes, a novel endo-β-1,4-glucanase (AfEG22), was purified 29.5-fold by gel
Juntao Sun et al.
Biotechnology letters, 33(11), 2193-2199 (2011-07-08)
A new fusion gene (Bgl-licMB), encoding β-1,3-1,4-glucanase both from Bacillus amyloliquefaciens (Bgl) and Clostridium thermocellum (licMB), was constructed via end-to-end fusion and expressed in Escherichia coli to improve hydrolytic activity and thermostability of β-1,3-1,4-glucanase. The results of enzymatic properties showed
Nhuan P Nghiem et al.
Applied biochemistry and biotechnology, 165(3-4), 870-882 (2011-06-15)
A fermentation process, which was designated the enhanced dry grind enzymatic (EDGE) process, has recently been developed for barley ethanol production. In the EDGE process, in addition to the enzymes normally required for starch hydrolysis, commercial β-glucanases were used to
Chengwei Hua et al.
Applied microbiology and biotechnology, 88(2), 509-518 (2010-07-21)
In this study, a novel beta-1,3-1,4-glucanase gene (designated as PtLic16A) from Paecilomyces thermophila was cloned and sequenced. PtLic16A has an open reading frame of 945 bp, encoding 314 amino acids. The deduced amino acid sequence shares the highest identity (61%)
C J O'Shea et al.
Journal of animal science, 88(4), 1411-1420 (2009-12-22)
The objective of the current experiment was to evaluate the influence of dietary cereal sources of beta(1,3)(1,4)-d-glucan (beta-glucan) and enzyme supplementation on indices of environmental pollution from finisher pigs. An experiment with a 2 x 2 factorial arrangement of treatments
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service