A4018
Agarose, low gelling temperature
Synonym(s):
2-Hydroxyethyl agarose
About This Item
Recommended Products
biological source
algae (marine)
Quality Level
form
powder
technique(s)
cell culture | mammalian: suitable
EEO
≤0.10
transition temp
congealing temperature 26-30 °C
gel strength
≥200 g/cm2 (1% gel)
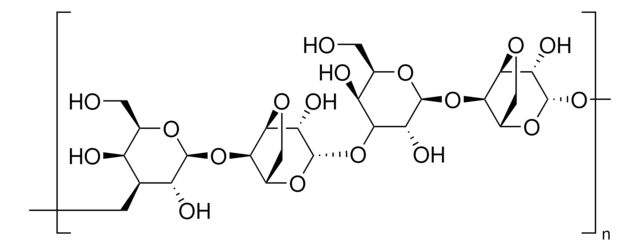
SMILES string
O1[C@H]([C@@H]([C@H]([C@H]([C@H]1CO)O)O[C@@H]4O[C@@H]5[C@H]([C@@H](OC5)[C@@H]4O)O[C@@H]6O[C@@H]([C@@H]([C@@H]([C@H]6O)O)O)CO)O)O[C@H]2[C@H]3OC[C@@H]2O[C@H]([C@H]3O)O
InChI
1S/C24H38O19/c25-1-5-9(27)11(29)12(30)22(38-5)41-17-8-4-36-20(17)15(33)24(40-8)43-18-10(28)6(2-26)39-23(14(18)32)42-16-7-3-35-19(16)13(31)21(34)37-7/h5-34H,1-4H2/t5-,6-,7+,8+,9+,10+,11+,12-,13+,14-,15+,16-,17-,18+,19+,20+,21-,22+,23+,24+/m1/s1
InChI key
MJQHZNBUODTQTK-WKGBVCLCSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
Agarose, low gelling temperature has been used:
- to immobilize zebra fish embryos during in vivo imaging experiments
- as a constituent of defined medium to culture bovine nucleus pulposus (NP) cells
- as a hydrogel, a scaffolding material for dental pulp regeneration
- in the form of pellets to determine the surface pH and conductivity of an acrylic emulsion film
Quality
Analysis Note
Sulfate content - used as an indicator of purity, since sulfate is the major ionic group present.
Gel strength - the force that must be applied to a gel to cause it to fracture.
Gel point - the temperature at which an aqueous agarose solution forms a gel as it cools. Agarose solutions exhibit hysteresis in the liquid-to-gel transition - that is, their gel point is not the same as their melting temperature.
Electroendosmosis (EEO) - a movement of liquid through the gel. Anionic groups in an agarose gel are affixed to the matrix and cannot move, but dissociable counter cations can migrate toward the cathode in the matrix, giving rise to EEO. Since electrophoretic movement of biopolymers is usually toward the anode, EEO can disrupt separations because of internal convection.
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
The Comet Assay, also called single cell gel electrophoresis (SCGE), is a sensitive and rapid technique for quantifying and analyzing DNA damage in individual cells.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service