217417
AmberChrom™ 1X8 Ion Exchange Resin
chloride form, 50-100 mesh
Synonym(s):
AmberChrom™ 1X8 chloride form, AmberChrom™ 1X8-100 ion exchange resin
About This Item
Recommended Products
Product Name
AmberChrom™ 1X8 50-100 MESH (CL-) Anion,
description
cross-linkage 8%
Quality Level
form
beads
crosslinking
8 % cross-linked
parameter
66 °C OH- form max. temp.
moisture
43-48%
technique(s)
LPLC: suitable
matrix
styrene-divinylbenzene (gel)
matrix active group
quaternary ammonium
particle size
50-100 mesh
operating pH
0-14
capacity
1.2 meq/mL by wetted bed volume
separation technique
anion exchange
SMILES string
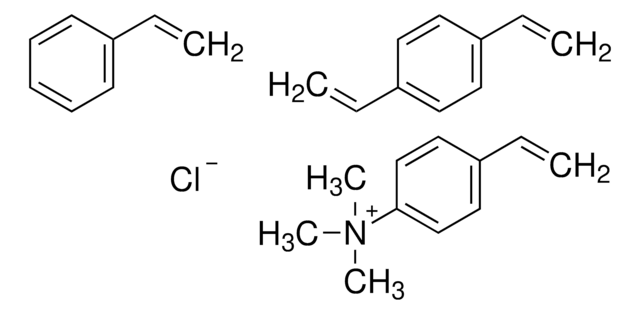
N(C)(C)C.C(C)c3c(cccc3)C=C.c2(c(cccc2)C=C)C=C.c1(ccccc1)C=C
InChI
1S/C10H12.C10H10.C8H8.C3H9N/c2*1-3-9-7-5-6-8-10(9)4-2;1-2-8-6-4-3-5-7-8;1-4(2)3/h3,5-8H,1,4H2,2H3;3-8H,1-2H2;2-7H,1H2;1-3H3
InChI key
HADXLRSCRPYPJJ-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
- to separate the 3H2O from D[2-3H] glucose using Dowex columns
- in a assay to determine the pantothenate kinase using Dowex column.
- to separate inositol phosphates (IPs) from myo-[3H]inositol by Dowex chromatography.
Features and Benefits
Other Notes
Legal Information
Storage Class
11 - Combustible Solids
wgk_germany
WGK 2
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Related Content
Low pressure liquid chromatography (LPLC) operates at low pressures, using a pump to drive the mobile phase onto the column.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service