SCC325
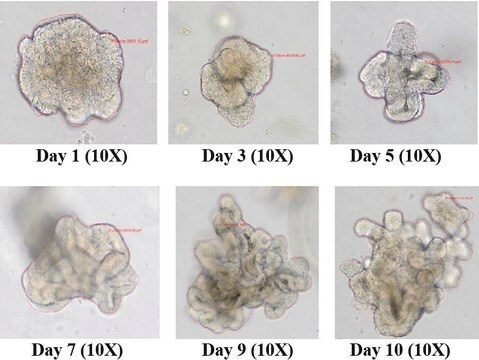
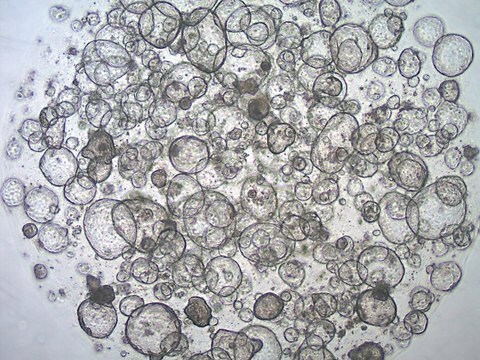
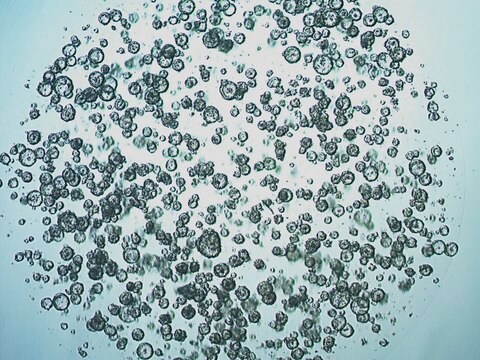
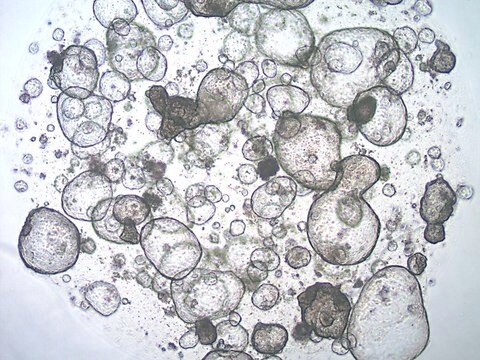
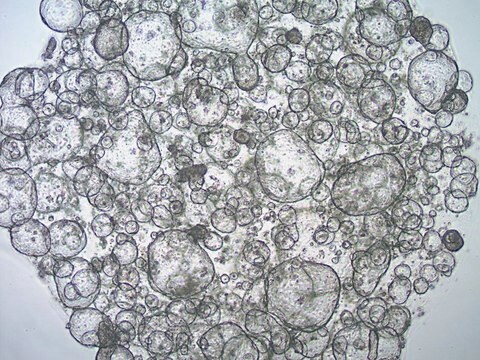
3dGRO® Colon Intestinal Organoids, Age 55 (Prep 89-C)
Cryopreserved patient derived gastrointestinal organoids (PDOs)
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
UNSPSC Code:
41106509
NACRES:
NA.81
Recommended Products
technique(s)
cell culture | stem cell: suitable
General description
Patient derived organoids (PDOs) are novel in vitro 3D cell models that preserve original tissue physiology and molecular pathology, thus representing a clinically-relevant alternative to traditional 2D cell lines and an effective tool to refine and reduce animal models. PDOs can be derived from adult patient biopsies or resected tissues containing native LGR5+ stem cell populations and cultured within an ECM-rich substrate using specialized organoid media such as L-WRN conditioned media. Adult tissue derived organoids are phenotypically and genetically stable in long term culture, presenting more mature phenotypes compared to iPSC-derived organoids. Importantly, PDOs have shown to be able to predict patient clinical responses to chemotherapeutics.
We are now offering a comprehensive biobank of highly characterized tissue-derived human gastrointestinal organoids from normal and diseased patients. The intestinal organoid biobank contains over 50 highly characterized intestinal organoids from both normal and diseased patients derived from multiple regions of the digestive systems including small intestine (duodenum, ileum), stomach, rectum and colon.
Cell Line Characteristics
We are now offering a comprehensive biobank of highly characterized tissue-derived human gastrointestinal organoids from normal and diseased patients. The intestinal organoid biobank contains over 50 highly characterized intestinal organoids from both normal and diseased patients derived from multiple regions of the digestive systems including small intestine (duodenum, ileum), stomach, rectum and colon.
Cell Line Characteristics
- Sex: Male
- Age: 55 Years
- Organ: Colon
- Disease: Normal
Cell Line Origin
Human, organoid
Packaging
≥1500 viable organoids/vial
Quality
- Viability: ≥1500 viable organoids/vial
- Organoid Growth: Pass
- Cells are tested negative for infectious diseases by a Human Essential CLEAR panel by Charles River Animal Diagnostic Services.
- Mycoplasma Contamination: Negative
- STR Profile: Pass
Storage and Stability
Store in liquid nitrogen. The organoids can be cultured for at least 10 passages after initial thawing without significantly affecting the cell marker expression and functionality.
Legal Information
3dGRO® organoids were derived utilizing HUB Organoid Technology
3dGRO is a registered trademark of Merck KGaA, Darmstadt, Germany
Disclaimer
RESEARCH USE ONLY. This product is regulated in France when intended to be used for scientific purposes, including for import and export activities (Article L 1211-1 paragraph 2 of the Public Health Code). The purchaser (i.e. enduser) is required to obtain an import authorization from the France Ministry of Research referred in the Article L1245-5-1 II. of Public Health Code. By ordering this product, you are confirming that you have obtained the proper import authorization.
Unless otherwise stated in our catalog or other company documentation accompanying the product(s), our products are intended for research use only and are not to be used for any other purpose, which includes but is not limited to, unauthorized commercial uses, in vitro diagnostic uses, ex vivo or in vivo therapeutic uses or any type of consumption or application to humans or animals. The purchaser of this product shall agree to HUB′s terms of use, which shall be separately acknowledged and accepted by such purchaser, prior to transfer of this Product to purchaser.
Storage Class
10 - Combustible liquids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service