E871
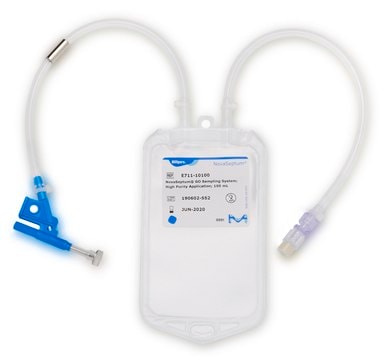
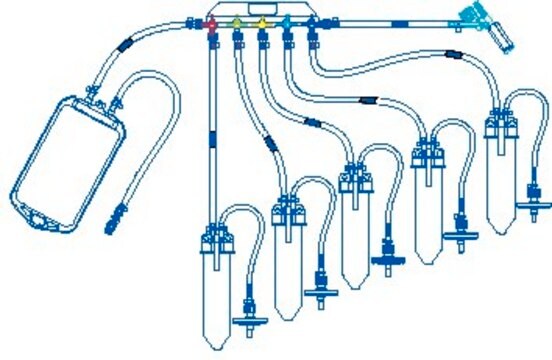
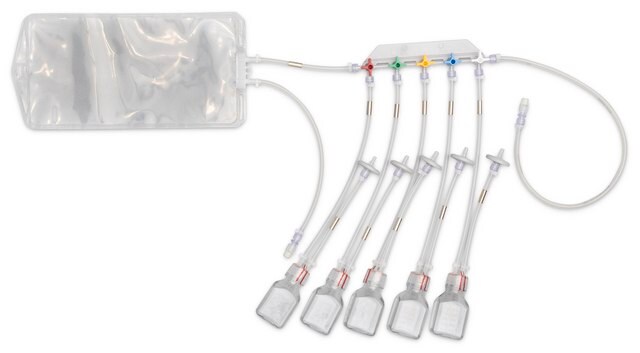
NovaSeptum® GO Bottle Sampling System
sterile; γ-irradiated
Synonym(s):
NovaSeptum® GO Bottle
About This Item
Recommended Products
material
316 stainless steel cannula (ASTM®)
PETG bottle
PureFlex™ polyethylene bottle
TPE tubing (Thermoplastic elastomer)
polyester body
silicone septum (platinum-cured)
Quality Level
agency
according to ISO 11137 (sterilization)
according to ISO 146441
meets USP 88 biological reactivity (all component materials)
sterility
sterile; γ-irradiated
product line
NovaSeptum® GO
feature
autoclavable: no
parameter
-80-50 °C temp. range (-112-122 °F)
0.50 bar max. pressure (7.25 psi)
impurities
<2.15 EU/device bacterial endotoxins (LAL test)
fitting
outlet (Cap)
application(s)
bioburden testing
cell therapy
gene therapy
life science and biopharma
mAb
ophthalmics
parenterals
pathogen testing
pharma/biopharma processes
sterile sampling
viral therapy
storage temp.
room temp
Looking for similar products? Visit Product Comparison Guide
General description
Application
Bioburden -
Archiving
Packaging
E871-80125 (125 ml bottle, 25 units per box)
E871-80250 (250 ml bottle, 20 units per box)
E871-80500 (500 ml bottle, 12 units per box)
Components
- Septum: Platinum-cured silicone
- Body: Polyester
- Cannula: ASTM® 316 L Stainless steel
- Fluid contact layer (bottle): Polyethylene Terephtalate Glycol(PETG)
- Tubing: Thermoplastic elastomer (TPE)
- Outlet Tubing: Cap
Preparation Note
Aqueous extraction contains < 2.15 EU per device as determined using the Limulus Amebocyte Lysate (LAL) test.
Legal Information
Not finding the right product?
Try our Product Selector Tool.
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service