06-933
Anti-acetyl-Lysine Antibody
serum, Upstate®
Synonym(s):
Acetylated Lysine Antibody
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
UNSPSC Code:
12352203
eCl@ss:
32160702
NACRES:
NA.41
Recommended Products
biological source
rabbit
Quality Level
antibody form
serum
antibody product type
primary antibodies
clone
polyclonal
species reactivity
vertebrates
manufacturer/tradename
Upstate®
technique(s)
immunoprecipitation (IP): suitable
western blot: suitable
isotype
IgG
shipped in
dry ice
target post-translational modification
acetylation (Lys)
Gene Information
human ... CECR2(27443)
General description
In the nucleus, DNA is tightly packed into nucleosomes generating an environment which is highly repressive towards DNA processes such as transcription. Acetylation of lysine residues within proteins has emerged as an important mechanism used by cells to overcome this repression. The acetylation of non-histone proteins such as transcription factors, as well as histones appears to be involved in this process. Acetylation may result in structural transitions as well as specific signaling within discrete chromatin domains. The role of acetylation in intracellular signaling has been inferred from the binding of acetylated peptides by the conserved bromodomain. Furthermore, recent findings suggest that bromodomain/acetylated-lysine recognition can serve as a regulatory mechanism in protein-protein interactions in numerous cellular processes such as chromatin remodeling and transcriptional activation.
Specificity
Acetyl-lysine containing proteins including histones, from sodium butyrate or Trichostatin A treated cells, p53 acetylated in vitro by p300 or PCAF, and chemically acetylated BSA.
Broad species reactivity expected.
Immunogen
A mixture of chemically acetylated antigens.
Application
Anti-acetyl-Lysine Antibody is a high quality Rabbit Polyclonal Antibody for the detection of acetyl-Lysine & has been validated in WB & IP.
Immunoprecipitation: 5 μL of a previous lot immunoprecipitated in vitro acetylated recombinant p300.
Western Blot Analysis: A 1:1000 dilution of the previous lot of the antibody detected:
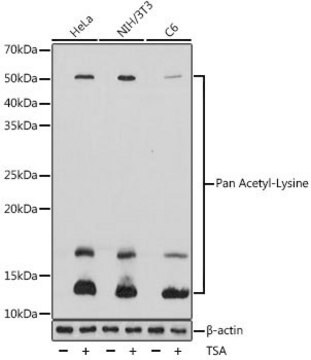
• Acetylated histones and to a lesser degree, other proteins in RIPA lysate from trichostatin A (5 μM) treated Cos-1 cells. Trichostatin A, an inhibitor of deacetylases also, was used to increase the level of acetylated histones. (Panel B).
• GST-p53 (100 ng) acetylated in vitro with either recombinant p300 or PCAF. (Panel C).
Western Blot Analysis: A 1:1000 dilution of the previous lot of the antibody detected:
• Acetylated histones and to a lesser degree, other proteins in RIPA lysate from trichostatin A (5 μM) treated Cos-1 cells. Trichostatin A, an inhibitor of deacetylases also, was used to increase the level of acetylated histones. (Panel B).
• GST-p53 (100 ng) acetylated in vitro with either recombinant p300 or PCAF. (Panel C).
Research Category
Epigenetics & Nuclear Function
Epigenetics & Nuclear Function
Research Sub Category
Chromatin Biology
Chromatin Biology
Quality
Evaluated by western blot on acid-extracted proteins from sodium butyrate treated HeLa cells, chemically acetylated BSA, acetylated histones and to a lesser degree, other proteins in trichostatin A treated Cos-1 cell lysate.
Western Blot Analysis: 1:1000-1:2000 dilution of this antibody detected:
• Acetylated histones in acid-extracted proteins from sodium butyrate (5 mM) treated HeLa cells. Sodium butyrate, an inhibitor of deacetylases, was used to increase the level of acetylated histones. (Panel A).
• Chemically acetylated BSA (10 ng) in a dot blot assay.
Western Blot Analysis: 1:1000-1:2000 dilution of this antibody detected:
• Acetylated histones in acid-extracted proteins from sodium butyrate (5 mM) treated HeLa cells. Sodium butyrate, an inhibitor of deacetylases, was used to increase the level of acetylated histones. (Panel A).
• Chemically acetylated BSA (10 ng) in a dot blot assay.
Target description
Dependent upon the molecular weight of the lysine acetylated protein being detected.
Physical form
Antiserum
Rabbit IgG serum containing 0.05% sodium azide and 30% glycerol.
Storage and Stability
Stable for 1 year at from date of receipt.
Analysis Note
Control
TSA-treated HeLa and NIH/3T3 cells, osteosarcoma tissue.
TSA-treated HeLa and NIH/3T3 cells, osteosarcoma tissue.
Other Notes
Concentration: Please refer to the Certificate of Analysis for the lot-specific concentration.
Legal Information
UPSTATE is a registered trademark of Merck KGaA, Darmstadt, Germany
Disclaimer
Unless otherwise stated in our catalog or other company documentation accompanying the product(s), our products are intended for research use only and are not to be used for any other purpose, which includes but is not limited to, unauthorized commercial uses, in vitro diagnostic uses, ex vivo or in vivo therapeutic uses or any type of consumption or application to humans or animals.
Not finding the right product?
Try our Product Selector Tool.
Storage Class
10 - Combustible liquids
wgk_germany
WGK 1
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
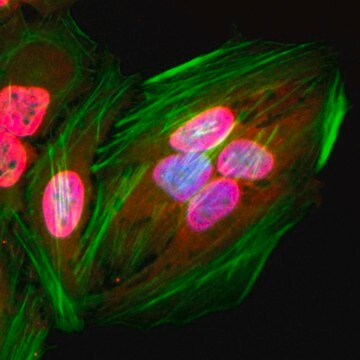
Cell cycle-dependent acetylation of Rb2/p130 in NIH3T3 cells.
Schwarze, F; Meraner, J; Lechner, M; Loidl, A; Stasyk, T; Laich, A; Loidl, P
Oncogene null
Identifying acetylated proteins in mitosis.
Carol Chuang,Li-Yuan Yu-Lee
Methods in Molecular Biology null
Restoration of senescent human diploid fibroblasts by modulation of the extracellular matrix.
Choi HR, Cho KA, Kang HT, Lee JB, Kaeberlein M, Suh Y, Chung IK, Park SC
Aging Cell null
Targeted acetylation of NF-kappaB/RelA and histones by epigenetic drugs reduces post-ischemic brain injury in mice with an extended therapeutic window.
Lanzillotta, Annamaria, et al.
Neurobiology of Disease, 49C, 177-189 (2012)
Multiple roles for acetylation in the interaction of p300 HAT with ATF-2.
Karanam, B; Wang, L; Wang, D; Liu, X; Marmorstein, R; Cotter, R; Cole, PA
Biochemistry null
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service