M31104
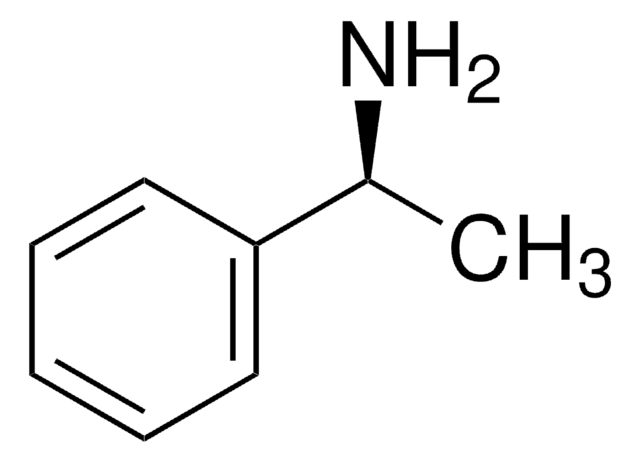
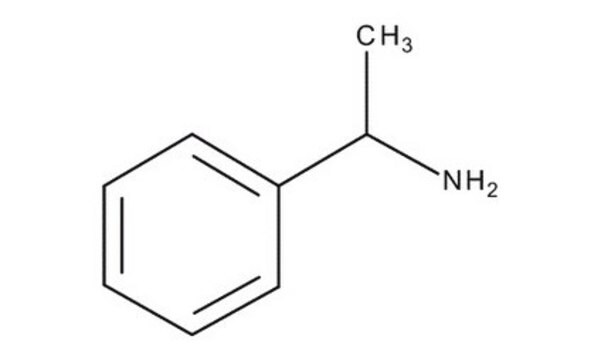
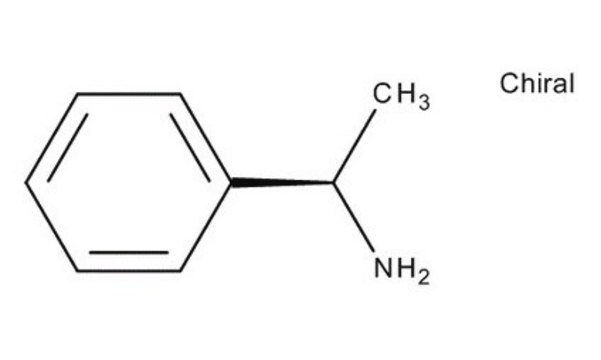
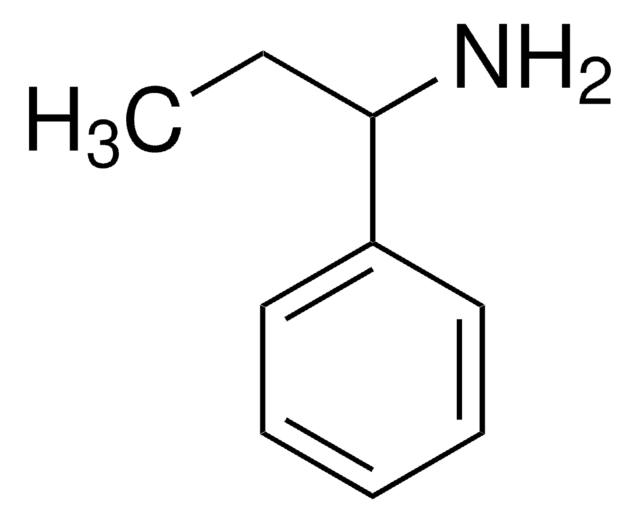
α-Methylbenzylamine
99%
Synonym(s):
(±)-α-Methylbenzylamine, alpha-Methylbenzylamine, (±)-1-Phenylethylamine
About This Item
Recommended Products
assay
99%
form
liquid
refractive index
n20/D 1.526 (lit.)
bp
185 °C/756 mmHg (lit.)
density
0.94 g/mL at 25 °C (lit.)
SMILES string
CC(N)c1ccccc1
InChI
1S/C8H11N/c1-7(9)8-5-3-2-4-6-8/h2-7H,9H2,1H3
InChI key
RQEUFEKYXDPUSK-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Oral - Eye Dam. 1 - Skin Corr. 1B
Storage Class
8A - Combustible corrosive hazardous materials
wgk_germany
WGK 1
flash_point_f
158.0 °F - closed cup
flash_point_c
70 °C - closed cup
ppe
Faceshields, Gloves, Goggles, type ABEK (EN14387) respirator filter
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Chromatograms
application for HPLCGlobal Trade Item Number
| SKU | GTIN |
|---|---|
| M31104-100ML | 4061834046363 |
| M31104-500ML | 4061836830991 |
| M31104-25ML | 4061834046370 |
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service