F408
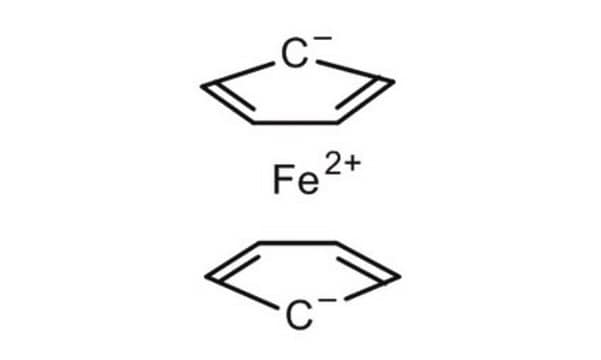
Ferrocene
98%
Synonym(s):
Bis(cyclopentadienyl)iron, Di(cyclopentadienyl)iron
About This Item
Recommended Products
vapor pressure
0.03 mmHg ( 40 °C)
Quality Level
assay
98%
reaction suitability
core: iron
reagent type: catalyst
bp
249 °C (lit.)
mp
172-174 °C (lit.)
λmax
358 nm
storage temp.
2-8°C
SMILES string
[Fe].[CH]1[CH][CH][CH][CH]1.[CH]2[CH][CH][CH][CH]2
InChI
1S/2C5H5.Fe/c2*1-2-4-5-3-1;/h2*1-5H;
InChI key
DFRHTHSZMBROSH-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
- As a scaffold for chiral ligands in asymmetric catalysis.
- In ferrocene-based electrolytes for dye sensitized solar cells applications.
- In the synthesis of unsymmetrical ferrocene ligands used as catalysts in cross-coupling, hydrogenation, allylic substitution, hydroformylation and aldol reactions.
- In the synthesis of two-dimensional hexagonally ordered mesoporous carbon (CMK-5) via chemical vapor deposition method, which is used as a catalyst support in oxidation of methanol.
- In the synthesis of molecular hybrids of ferrocene and fullerene (Bucky ferrocenes).
- In the preparation of ferrocenium salt which is used as a mild oxidant.
signalword
Danger
Hazard Classifications
Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Aquatic Chronic 1 - Flam. Sol. 1 - Repr. 1B - STOT RE 2 Inhalation
target_organs
Liver
Storage Class
4.1B - Flammable solid hazardous materials
wgk_germany
WGK 2
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type P3 (EN 143) respirator cartridges
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Ferrocenylmethyl Methacrylate for Functional Polymers
Atomic layer deposition (ALD) techniques have emerged in the last ten years to meet various needs including semiconductor device miniaturization, conformal deposition on porous structures and coating of nanoparticles. ALD is based on two sequential self-limiting surface reactions.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service