All Photos(1)
About This Item
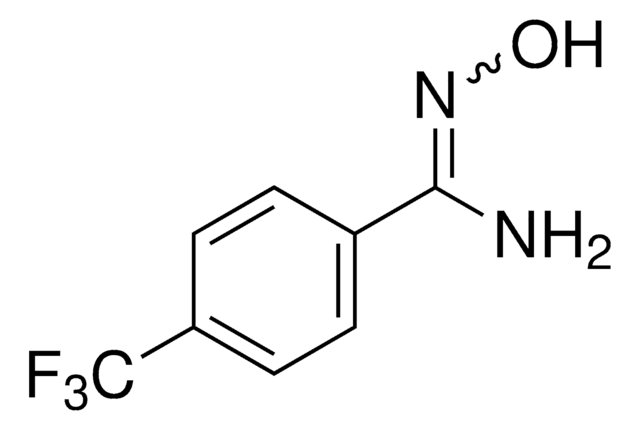
Linear Formula:
CF3OC6H4C(=NOH)NH2
CAS Number:
Molecular Weight:
220.15
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
assay
97%
mp
113-115 °C (lit.)
functional group
amine
fluoro
oxime
SMILES string
N\C(=N/O)c1ccc(OC(F)(F)F)cc1
InChI
1S/C8H7F3N2O2/c9-8(10,11)15-6-3-1-5(2-4-6)7(12)13-14/h1-4,14H,(H2,12,13)
InChI key
COHKFOZYLCDVRK-UHFFFAOYSA-N
General description
4-(Trifluoromethoxy)benzamidoxime (4-TFMBAO) is a benzamidoxime (BAO) derivative containing amidoxime functional group. Its density and freezing point have been determined.
Application
4-(Trifluoromethoxy)benzamidoxime (4-TFMBAO) is suitable reactant in the fluorescence (FL) deriving reaction, one of the widely-used methodology specifically used to quantify uracil. It may be used as a reactant in the synthesis of oxadiazoles. It may also be used a fluorogenic agent in the quantification of orotic acid by spectrofluorometric method in human biological specimens.
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Sensitive and Selective Determination of Orotic Acid in Biological Specimens Using a Novel Fluorogenic Reaction.
Yin S, et al.
Journal of Fluorescence, 25(4), 1005-1011 (2015)
Thomas E Barta et al.
Bioorganic & medicinal chemistry letters, 21(10), 2820-2822 (2011-04-22)
Seeking compounds preferentially potent and selective for MMP-13, we reported in the preceding Letter on a series of hydroxamic acids with a flexible benzamide tail groups.(1a) Here, we replace the amide moiety with non-hydrolyzable heterocycles in an effort to improve
Yaws CL.
The Yaws Handbook of Physical Properties for Hydrocarbons and Chemicals, 185-185 (2015)
Evan R Abt et al.
Cell chemical biology, 27(2), 197-205 (2019-11-18)
Biosynthesis of the pyrimidine nucleotide uridine monophosphate (UMP) is essential for cell proliferation and is achieved by the activity of convergent de novo and salvage metabolic pathways. Here we report the development and application of a cell-based metabolic modifier screening
Takayuki Shibata et al.
Analytica chimica acta, 674(2), 234-238 (2010-08-04)
Facile and specific methods to quantify a nucleobase in biological samples are of great importance for diagnosing disorders in nucleic acid metabolism. In the present study, a novel fluorogenic reaction specific for uracil has been developed. The reaction was carried
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service