40718
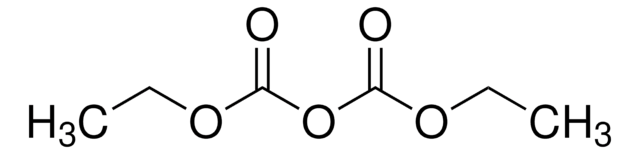
Diethyl pyrocarbonate
96% (NT)
Synonym(s):
DEP, DEPC, Diethyl dicarbonate, Diethyl oxydiformate, Ethoxyformic acid anhydride
About This Item
Recommended Products
Quality Level
assay
96% (NT)
form
liquid
color
APHA: ≤20
refractive index
n20/D 1.398 (lit.)
n20/D 1.398
bp
93-94 °C/18 mmHg (lit.)
solubility
95% ethanol: 4.5 g/10 mL, clear, colorless
density
1.121 g/mL at 20 °C
1.101 g/mL at 25 °C (lit.)
shipped in
wet ice
storage temp.
2-8°C
SMILES string
CCOC(=O)OC(=O)OCC
InChI
1S/C6H10O5/c1-3-9-5(7)11-6(8)10-4-2/h3-4H2,1-2H3
InChI key
FFYPMLJYZAEMQB-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
- For the modification of histidyl residues in proteins.
- As a nuclease inhibitor, for the extraction of undegraded nucleic acids from etiolated and green plant tissues.
- For the modification of linear and supercoiled plasmid DNAs.
- As a chemical probe to investigate the secondary structure in negatively supercoiled DNA.
- For cabethoxylation of histidine residues of actin.
Biochem/physiol Actions
signalword
Warning
hcodes
Hazard Classifications
Acute Tox. 4 Oral
Storage Class
10 - Combustible liquids
wgk_germany
WGK 3
flash_point_f
156.2 °F - closed cup
flash_point_c
69 °C - closed cup
ppe
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service