推薦產品
產品名稱
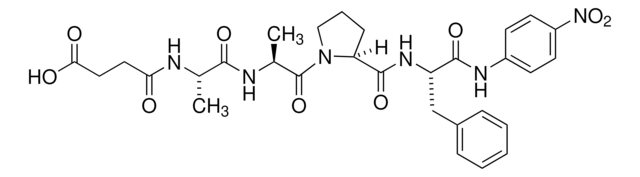
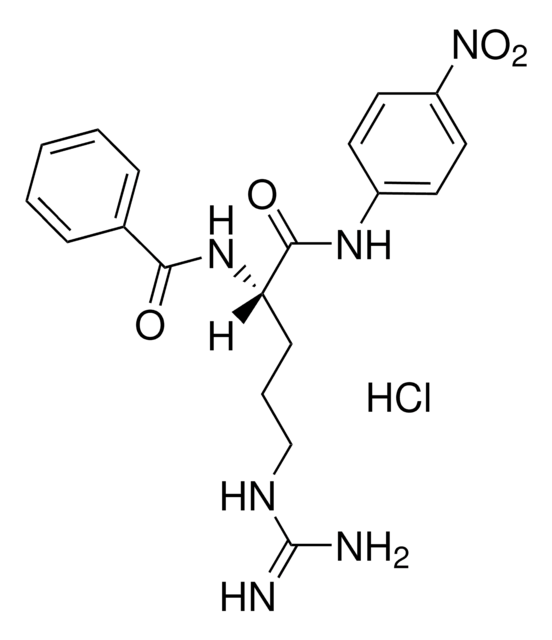
N-琥珀酰- L -苯丙氨酸- p -硝基苯胺, protease substrate
化驗
≥98% (TLC)
形狀
powder
溶解度
dioxane: 25 mg/mL, clear, colorless to faintly yellow
儲存溫度
−20°C
SMILES 字串
OC(=O)CCC(=O)N[C@@H](Cc1ccccc1)C(=O)Nc2ccc(cc2)N(=O)=O
InChI
1S/C19H19N3O6/c23-17(10-11-18(24)25)21-16(12-13-4-2-1-3-5-13)19(26)20-14-6-8-15(9-7-14)22(27)28/h1-9,16H,10-12H2,(H,20,26)(H,21,23)(H,24,25)/t16-/m0/s1
InChI 密鑰
KNBLWBFJHZHYFG-INIZCTEOSA-N
應用
N -琥珀酰- L -苯丙氨酸- p -硝基苯胺已被用作糜蛋白酶水解活性的底物和摩尔吸光实验。也用于体外监测 α-糜蛋白酶抑制试验。
生化/生理作用
糜蛋白酶水解活性的底物 N -琥珀酰- L -苯丙氨酸- p -硝基苯胺形成黄色生色团 p -硝基苯胺,可在 410 nm 处分光光度法测定。
基底
据报道,这是一种糜蛋白酶的底物。
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
Eyeshields, Gloves, type N95 (US)
客戶也查看了
Isabel Pastor et al.
The journal of physical chemistry. B, 115(5), 1115-1121 (2010-12-31)
Traditionally, studies on the diffusion-controlled reaction of biological macromolecules have been carried out in dilute solutions (in vitro). However, in an intracellular environment (in vivo), there is a high concentration of macromolecules, which results in nonspecific interactions (macromolecular crowding). This
E Iu Maksareva et al.
Bioorganicheskaia khimiia, 21(1), 24-27 (1995-01-01)
Subtilisin Carlsberg (E.C. 3.4.21.14) catalyzes the hydrolysis of N-succinyl-L-phenylalanine p-nitroanilide in solid-state solvent-free hydrated protein-substrate mixtures. This process needs a certain critical degree of hydration of the protein molecule which is attained at the relative water vapour pressure (p/ps) above
Mercury (II) binds to both of chymotrypsin's histidines, causing inhibition followed by irreversible denaturation/aggregation
Stratton A, et al.
Protein Science, 26(2), 292-305 (2017)
V V Mozhaev et al.
Biochemistry and molecular biology international, 34(1), 191-199 (1994-08-01)
Biocatalytic transformations in reversed micelles formed by anionic surfactant Aerosol OT in octane have been studied at high pressures by an example of alpha-chymotrypsin-catalyzed hydrolysis of N-carbobenzoxy-L-tyrosine p-nitrophenyl ester and N-succinyl-L-phenylalanine p-nitroanilide. For the first time it has been found
[ON THE DETERMINATION OF TRYPSIN AND CHYMOTRYPSIN WITH AMINO-P-NITROANILIDES].
W NAGEL et al.
Hoppe-Seyler's Zeitschrift fur physiologische Chemie, 340, 1-10 (1965-01-01)
文章
Analytical Enzyme Chymotrypsin: Chymotrypsin is produced in the acinar cells of the pancreas as the inactive precursor, chymotrypsinogen.
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務