A8126
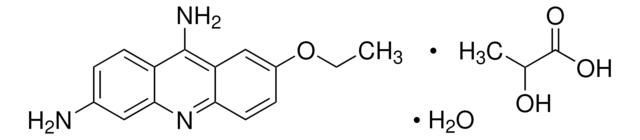
Acriflavine
Powder
Synonym(s):
3,6-Diamino-10-methylacridinium chloride mixt. with 3,6-diaminoacridine (proflavine), Euflavine, Trypaflavine Neutral
About This Item
Recommended Products
product name
Acriflavine, fluorescent label
Quality Level
form
powder
mp
179-181 °C
solubility
H2O: 0.33 g/mL (lit.)(lit.)
εmax
≥48000 at 459-465 nm in methanol at 0.004 g/L
≥50000 at 259-265 nm in methanol at 0.004 g/L
application(s)
diagnostic assay manufacturing
hematology
histology
storage temp.
room temp
SMILES string
[Cl-].Nc1ccc2cc3ccc(N)cc3nc2c1.C[n+]4c5cc(N)ccc5cc6ccc(N)cc46
InChI
1S/C14H13N3.C13H11N3.ClH/c1-17-13-7-11(15)4-2-9(13)6-10-3-5-12(16)8-14(10)17;14-10-3-1-8-5-9-2-4-11(15)7-13(9)16-12(8)6-10;/h2-8H,1H3,(H3,15,16);1-7H,14-15H2;1H
InChI key
PEJLNXHANOHNSU-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
Excitation and emission wavelengths in various solvents :
- Methanol: λex = 424 nm; λem = 518 nm
- Ethanol: λex = 426 nm; λem = 524 nm
- Propanol: λex = 430 nm; λem = 512 nm
- Butanol: λex = 430 nm; λem = 526 nm
- Formamide: λex = 434 nm; λem = 524 nm
- Glycerol: λex = 432 nm; λem = 540 nm
- Water: λex = 416 nm; λem = 514 nm
Insoluble in ether, chloroform, and fixed oils. Utilized in fluorescence steady state measurements as a donor molecule (when paired with rhodamine 6G as the acceptor) to function as a pH sensor .
- Acriflavine has been used in the agglutination test to distinguish between smooth and rough colony formation of Brucella melitensis.
- It has been used as an Ago2 (argonaute 2) inhibitor.
- It has been used as an inhibitor of HIF-ARNT (hypoxia-inducible factor - aryl hydrocarbon receptor nuclear translocator) complex formation.
- It has been used to study the bacteriocin production by Carnobacterium piscicola.
Biochem/physiol Actions
signalword
Warning
hcodes
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service