15450
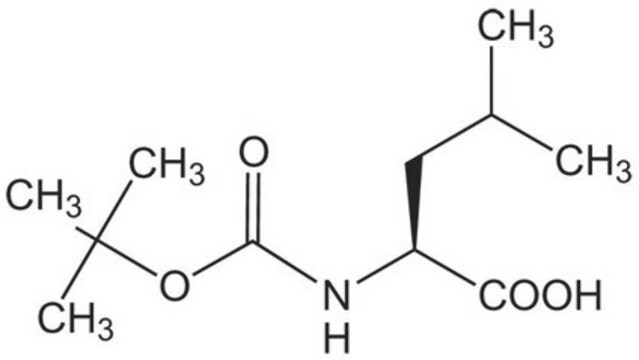
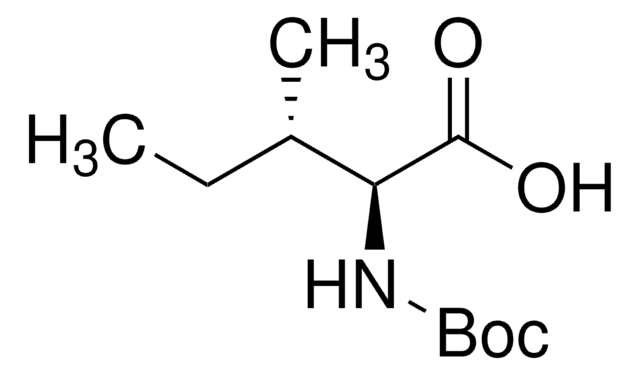
Boc-Leu-OH hydrate
≥99.0% (HPLC)
Synonym(s):
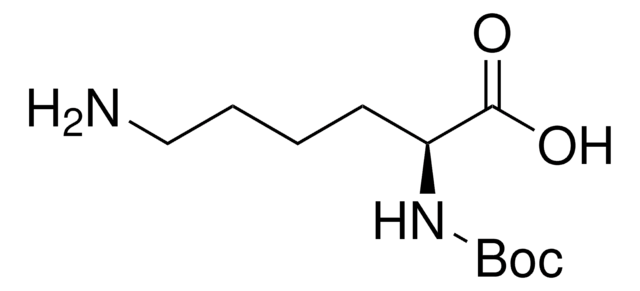
Boc-L-leucine
About This Item
Recommended Products
Quality Level
assay
≥99.0% (HPLC)
form
solid
optical activity
[α]20/D −25±0.5°, c = 2% in acetic acid
reaction suitability
reaction type: Boc solid-phase peptide synthesis
reaction type: C-H Activation
reagent type: ligand
reaction type: Peptide Synthesis
mp
85-90 °C
application(s)
peptide synthesis
functional group
amine
carboxylic acid
SMILES string
CC(C)C[C@H](NC(OC(C)(C)C)=O)C(O)=O
Application
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Articles
With a growing peptide drug market the fast, reliable and uncomplicated synthesis of peptides is of paramount importance.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service