206555
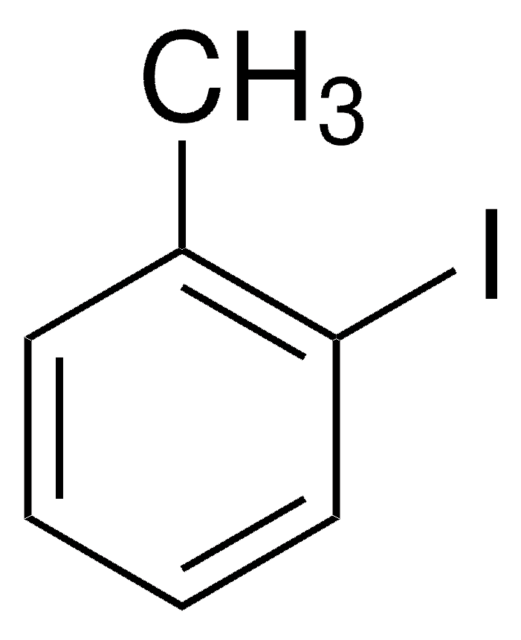
4-Iodotoluene
99%
Synonym(s):
1-Iodo-4-methylbenzene
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Linear Formula:
CH3C6H4I
CAS Number:
Molecular Weight:
218.03
Beilstein/REAXYS Number:
1903637
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
assay
99%
form
solid
bp
211.5 °C (lit.)
mp
33-35 °C (lit.)
SMILES string
Cc1ccc(I)cc1
InChI
1S/C7H7I/c1-6-2-4-7(8)5-3-6/h2-5H,1H3
InChI key
UDHAWRUAECEBHC-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
4-Iodotoluene undergoes Suzuki-Miyaura coupling reaction with phenylboronic acid catalyzed by (Ni,Mg)3Si2O5(OH)4 solid-solution nanotubes loaded with palladium. Cobalt-catalyzed coupling of 4-iodotoluene with thiophenols and alkanethiols has been investigated. Palladium/copper-catalyzed Sonogashira cross-coupling reaction of 4-iodotoluene with phenylacetylene has been studied.
signalword
Warning
hcodes
Hazard Classifications
Acute Tox. 4 Oral - Skin Irrit. 2
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
194.0 °F - closed cup
flash_point_c
90 °C - closed cup
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Wancheng Zhu et al.
Inorganic chemistry, 51(11), 6020-6031 (2012-05-16)
(Ni(1-x),Mg(x))(3)Si(2)O(5)(OH)(4) solid-solution nanotubes (NTs) with tunable compositions were hydrothermally synthesized by altering the molar ratio of Mg(2+) to Ni(2+). The as-synthesized NTs were loaded with sub-0.06 wt % palladium (Pd; ∼0.045 wt %) for Suzuki-Miyaura (SM) coupling reactions between iodobenzene
Takashi Mino et al.
The Journal of organic chemistry, 71(25), 9499-9502 (2006-12-02)
Palladium/copper-catalyzed Sonogashira cross-coupling reaction of aryl halides with a variety of terminal alkynes under amine-free conditions in dimethylformamide (DMF) at 80 degrees C gave internal arylated alkynes using PdCl2(MeCN)2 with phosphine-free hydrazone 2a as a ligand and CuI as the
Ying-Chieh Wong et al.
Organic letters, 8(24), 5613-5616 (2006-11-17)
A new cobalt-catalyzed coupling of aryl halides with thiophenols and alkanethiols is reported. A variety of aryl sulfides can be prepared in excellent yields under mild reaction conditions using 1-2 mol % of CoI2(dppe) and Zn. This new cobalt-catalyzed coupling
V Kolaříková et al.
Dalton transactions (Cambridge, England : 2003), 44(45), 19663-19673 (2015-09-17)
Using three different approaches, racemic 1-(perfluoroalkyl)ethylamines were synthesized from perfluoroalkyl iodides or perfluoroalkanoic acids, and further transformed to the corresponding N,N'-disubstituted ethane-1,2-diimines and ethane-1,2-diamines as mixtures of diastereoisomers. Their cyclization afforded imidazolium or dihydroimidazolium salts, which led to silver or
Zengyan Wei et al.
Nature communications, 5, 3870-3870 (2014-05-16)
The shape-controlled synthesis of nanoparticles was established in single-phase solutions by controlling growth directions of crystalline facets on seed nanocrystals kinetically; however, it was difficult to rationally predict and design nanoparticle shapes. Here we introduce a methodology to fabricate nanoparticles
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service