추천 제품
Quality Level
분석
≥85.0% (HPLC)
저장 온도
−20°C
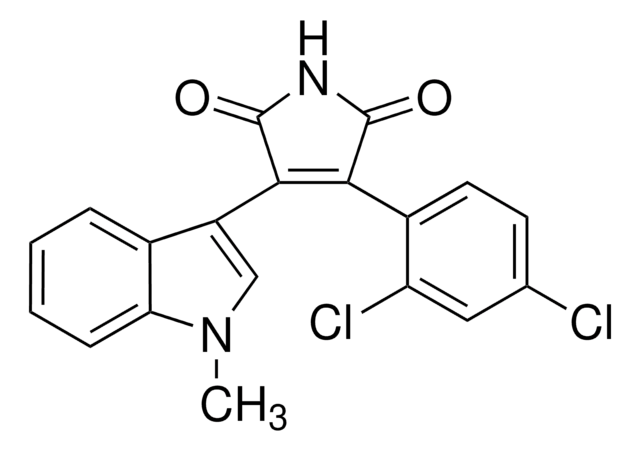
SMILES string
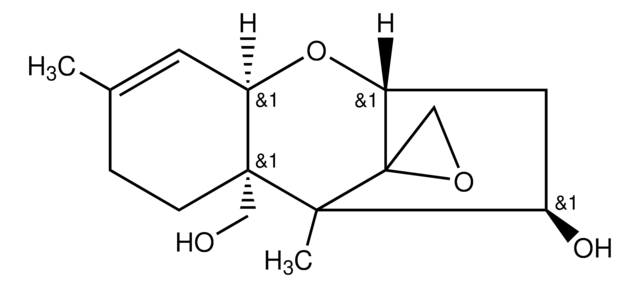
CC1CCOC(=O)\C=C\C=C\C(=O)OC2CC3OC4C=C(C)CCC4(COC(=O)C1O)C2(C)C35CO5
InChI
1S/C27H34O9/c1-16-8-10-26-14-33-24(31)23(30)17(2)9-11-32-21(28)6-4-5-7-22(29)36-18-13-20(35-19(26)12-16)27(15-34-27)25(18,26)3/h4-7,12,17-20,23,30H,8-11,13-15H2,1-3H3/b6-4+,7-5+
InChI key
NLUGUZJQJYVUHS-YDFGWWAZSA-N
일반 설명
Verrucarin derived from the Myrothecium verrucari, a fungal plant pathogen is a macrocyclic trichothecene compound.
애플리케이션
Verrucarin A from Myrothecium sp has been used:
- as a translation initiation inhibitor in P. falciparum W2 strain
- as a peptidyl transfer inhibitor to test sensitivity in methyltransferase knock-out S. cerevisiae cells
- to test its cytotoxicity in renal cell carcinoma cells
생화학적/생리학적 작용
Verrucarin A binds to the P and A sites in ribosomes and blocks peptide-bond and translation initiation. It inhibits the clear cell renal cell carcinoma (CCRCC) proliferation. Verrucarin A blocks the peptidyl transferase activity and favors apoptosis induction in cancer cells
Verrucarin A is phytotoxic to plantlet cultures and cytotoxic to cultured mammalian cell lines.
기타 정보
Macrocyclic trichothecene mycotoxin.
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 1 Inhalation - Acute Tox. 2 Dermal - Acute Tox. 2 Oral
Storage Class Code
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
WGK
WGK 3
개인 보호 장비
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
이미 열람한 고객
Chien-Shan Cheng et al.
Cancer cell international, 19, 92-92 (2019-04-24)
In the past decade, increasing research attention investigated the novel therapeutic potential of steroidal cardiac glycosides in cancer treatment. Huachansu and its main active constituent Bufalin have been studied in vitro, in vivo and clinical studies. This review aims to
Antonio L Amelio et al.
Proceedings of the National Academy of Sciences of the United States of America, 104(51), 20314-20319 (2007-12-14)
Signal transduction pathways often use a transcriptional component to mediate adaptive cellular responses. Coactivator proteins function prominently in these pathways as the conduit to the basic transcriptional machinery. Here we present a high-throughput cell-based screening strategy, termed the "coactivator trap,"
Charles S Bond et al.
The Journal of cell biology, 186(5), 637-644 (2009-09-02)
Paraspeckles are ribonucleoprotein bodies found in the interchromatin space of mammalian cell nuclei. These structures play a role in regulating the expression of certain genes in differentiated cells by nuclear retention of RNA. The core paraspeckle proteins (PSF/SFPQ, P54NRB/NONO, and
Mukhtar Jaderson et al.
Safety and health at work, 10(2), 196-204 (2019-07-13)
Liquid chromatography-tandem mass spectrometry (LC-MSMS) for simultaneous analysis of multiple microbial secondary metabolites (MSMs) is potentially subject to interference by matrix components. We examined potential matrix effects (MEs) in analyses of 31 MSMs using ultraperformance LC-MSMS. Twenty-one dust aliquots from three
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.