모든 사진(1)
About This Item
실험식(Hill 표기법):
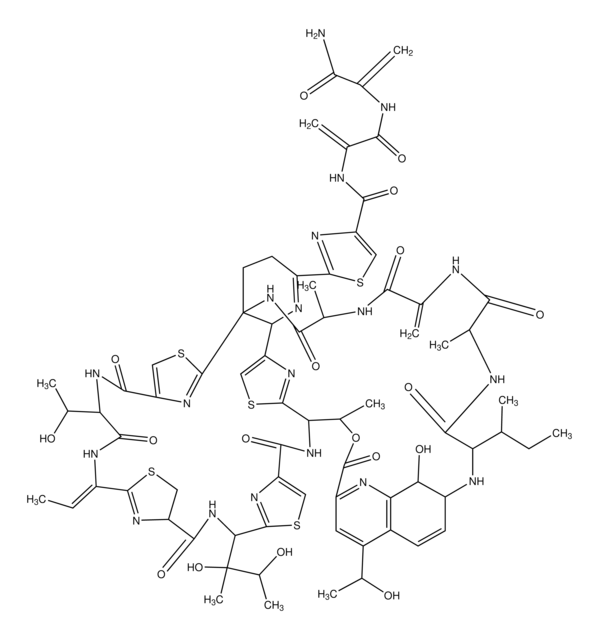
C30H28N6O6S4
CAS Number:
Molecular Weight:
696.84
MDL number:
UNSPSC 코드:
12352202
PubChem Substance ID:
NACRES:
NA.77
추천 제품
Quality Level
분석
≥95% (HPLC)
양식
powder
저장 온도
2-8°C
SMILES string
CN1C(=O)C23CC4(C(Nc5ccccc45)N2C(=O)C1(CO)SS3)C67CC89SSC(CO)(N(C)C8=O)C(=O)N9C6Nc%10ccccc7%10
InChI
1S/C30H28N6O6S4/c1-33-21(39)27-11-25(15-7-3-5-9-17(15)31-19(25)35(27)23(41)29(33,13-37)45-43-27)26-12-28-22(40)34(2)30(14-38,46-44-28)24(42)36(28)20(26)32-18-10-6-4-8-16(18)26/h3-10,19-20,31-32,37-38H,11-14H2,1-2H3
InChI key
PZPPOCZWRGNKIR-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
Chaetocin is a fungal metabolite with antimicrobial and cytostatic activity. It belongs to the 3,6-epidithio-diketopiperazines class of which gliotoxin, sporidesmin, aranotin, oryzachloride, verticillin A and the melinacidins are members. Chaetocin is a molecular dimer of two five-membered rings cis fused.
애플리케이션
Chaetocin from Chaetomium minutum has been used to determine its effects on sensitization of various cells. It has also been used to determine the biological functions of OS-induced heterochromatin formation.
생화학적/생리학적 작용
Chaetocin is a competitive inhibitor for S-adenosylmethionine. The specificity of chaetocin for SU(VAR)3-9 makes this compound an excellent tool for the study of heterochromatin-mediated gene repression.
Chaetocin is an antibacterial mycotoxin. It is a specific inhibitor of lysine-specific histone methyltransferase making the compound useful in the study of heterochromatin mediated gene repression. Chaetocin is a selective competitive inhibitor of thioredoxin reductase-1 (TrxR1), a putative mechanism of its anticancer activity.
Methyltransferase making the compound useful in the study of heterochromatin mediated gene repression.
제조 메모
Soluble in DMSO. At 1 mg/ml the solution is stable for 1 week at 2-8 °C.
신호어
Warning
유해 및 위험 성명서
예방조치 성명서
Hazard Classifications
Acute Tox. 4 Oral
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
dust mask type N95 (US), Eyeshields, Gloves
이미 열람한 고객
The epipolythiodioxopiperazine (ETP) class of fungal toxins: distribution, mode of action, functions and biosynthesis
Gardiner D M, et al.
Microbiology, 151(4), 1021-1032 (2005)
Jennifer D Tibodeau et al.
Antioxidants & redox signaling, 11(5), 1097-1106 (2008-11-13)
We recently reported that the antineoplastic thiodioxopiperazine natural product chaetocin potently induces cellular oxidative stress, thus selectively killing cancer cells. In pursuit of underlying molecular mechanisms, we now report that chaetocin is a competitive and selective substrate for the oxidative
Justin Kim et al.
Journal of the American Chemical Society, 132(41), 14376-14378 (2010-09-28)
A highly stereoselective and systematic strategy for the introduction of polysulfides in the synthesis of epipolythiodiketopiperazines is described. We report the first total synthesis of dimeric epitri- and epitetrathiodiketopiperazines.
Identification of a specific inhibitor of the histone methyltransferase SU (VAR) 3-9
Greiner D, et al.
Nature Chemical Biology, 1(3), 143-143 (2005)
Yoon-Mi Lee et al.
Hepatology (Baltimore, Md.), 53(1), 171-180 (2010-12-09)
Chaetocin, an antibiotic produced by Chaetomium species fungi, was recently found to have antimyeloma activity. Here we examined whether chaetocin has anticancer activities against solid tumors. Chaetocin inhibited the growth of mouse and human hepatoma grafts in nude mice. Immunohistochemical
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.