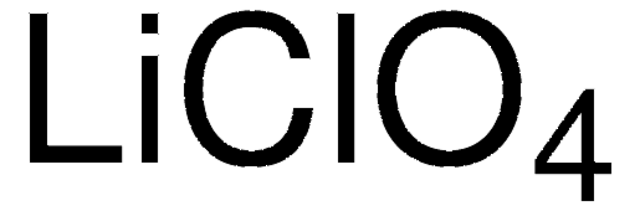62580
Lithium perchlorate
purum p.a., ≥98.0% (calc. based on dry substance, T), powder
로그인조직 및 계약 가격 보기
모든 사진(1)
About This Item
Linear Formula:
LiClO4
CAS Number:
Molecular Weight:
106.39
EC Number:
MDL number:
UNSPSC 코드:
12352302
PubChem Substance ID:
NACRES:
NA.21
solubility:
alcohol: soluble(lit.)
water: soluble(lit.)
water: soluble(lit.)
추천 제품
grade
purum p.a.
분석
≥98.0% (calc. based on dry substance, T)
양식
powder
반응 적합성
reagent type: oxidant
불순물
≤3% water
pH
6.0-9.5 (25 °C, 106.4 g/L)
mp
236 °C (lit.)
solubility
alcohol: soluble(lit.)
water: soluble(lit.)
음이온 미량물
chloride (Cl-): ≤50 mg/kg
sulfate (SO42-): ≤200 mg/kg
양이온 미량물
Ca: ≤100 mg/kg
Cd: ≤50 mg/kg
Co: ≤50 mg/kg
Cu: ≤50 mg/kg
Fe: ≤50 mg/kg
K: ≤500 mg/kg
Na: ≤100 mg/kg
Ni: ≤50 mg/kg
Pb: ≤50 mg/kg
Zn: ≤50 mg/kg
SMILES string
[Li+].[O-]Cl(=O)(=O)=O
InChI
1S/ClHO4.Li/c2-1(3,4)5;/h(H,2,3,4,5);/q;+1/p-1
InChI key
MHCFAGZWMAWTNR-UHFFFAOYSA-M
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
Lithium perchlorate (LiClO4) is a colorless lithium salt. Upon crystallization from its aqueous solution, it affords lithium perchlorate trihydrate (LiClO4.3H2O). It can be synthesized by reacting lithium chloride with perchloric acid.
애플리케이션
Lithium perchlorate (LiClO4) may be used as a catalyst in the synthesis of bis(indolyl)methanes and in the Baylis-Hillman coupling reaction of α,β-unsaturated carbonyl compounds with aldehydes.
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 4 Oral - Eye Dam. 1 - Ox. Sol. 2 - Skin Corr. 1A - STOT SE 3
표적 기관
Respiratory system
Storage Class Code
5.1A - Strongly oxidizing hazardous materials
WGK
WGK 1
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
Lithium perchlorate-accelerated Baylis-Hillman reactions.
Kawamura M and Kobayashi S.
Tetrahedron Letters, 40(8), 1539-1542 (1999)
Lithium perchlorate catalyzed reactions of indoles: An expeditious synthesis of bis (indolyl) methanes.
Yadav JS, et al.
Synthesis, 2001(5), 0783-0787 (2001)
Shokaku Kim et al.
Organic letters, 4(21), 3735-3737 (2002-10-12)
[reaction: see text] N-Acyliminium cation of prolines was efficiently generated to accumulate in an undivided cell at 0 degrees C by an anodic oxidation of N-acylprolines or alpha'-phenylsulfanylated N-acylproline derivatives in a lithium perchlorate/nitromethane solution. The iminium cation intermediates gave
L H Sim et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 76(3-4), 287-292 (2010-05-07)
The interaction behaviours between components of polyacrylate (PAc)/poly(ethylene oxide) (PEO) and lithium perchlorate (LiClO(4)) were investigated in detail by Attenuated Total Reflectance (ATR)-Fourier Transformed Infrared (FTIR) spectroscopy. Solution cast films of the PAc/PEO and PAc/PEO/LiClO(4) were examined. No obvious shifting
Natalia Varaksa et al.
Proceedings of the National Academy of Sciences of the United States of America, 99(8), 5012-5017 (2002-04-18)
The adsorption of the trigonal connector, 1,3,5-tris[10-(3-ethylthiopropyl)dimethylsilyl-1,10-dicarba-closo-decaboran-1-yl]benzene (1), from acetonitrile/0.1 M LiClO(4) on the surface of mercury at potentials ranging from +0.3 to -1.4 V (vs. aqueous Ag/AgCl/1 M LiCl) was examined by voltammetry, Langmuir isotherms at controlled potentials, and
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.