Y0001162
Misoprostol for system suitability
European Pharmacopoeia (EP) Reference Standard
동의어(들):
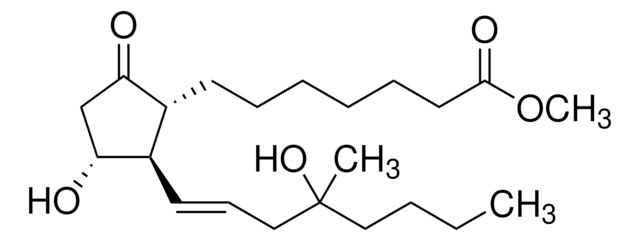
Misoprostol, (±)-15-Deoxy-(16RS)-16-hydroxy-16-methylprostaglandin E1 methyl ester
로그인조직 및 계약 가격 보기
모든 사진(1)
About This Item
실험식(Hill 표기법):
C22H38O5
CAS Number:
Molecular Weight:
382.53
MDL number:
UNSPSC 코드:
41116107
PubChem Substance ID:
NACRES:
NA.24
추천 제품
Grade
pharmaceutical primary standard
API family
misoprostol
제조업체/상표
EDQM
응용 분야
pharmaceutical (small molecule)
형식
neat
저장 온도
−20°C
SMILES string
CCCCC(C)(O)C\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(=O)OC
InChI
1S/C22H38O5/c1-4-5-14-22(2,26)15-10-12-18-17(19(23)16-20(18)24)11-8-6-7-9-13-21(25)27-3/h10,12,17-18,20,24,26H,4-9,11,13-16H2,1-3H3/b12-10+/t17-,18-,20-,22?/m1/s1
InChI key
OJLOPKGSLYJEMD-URPKTTJQSA-N
유전자 정보
human ... PTGER3(5733)
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
This product is provided as delivered and specified by the issuing Pharmacopoeia. All information provided in support of this product, including SDS and any product information leaflets have been developed and issued under the Authority of the Issuing Pharmacopoeia. For further information and support please go to the website of the issuing Pharmacopoeia.
애플리케이션
Misoprostol for system suitability EP Reference standard, intended for use in laboratory tests only as specifically prescribed in the European Pharmacopoeia.
생화학적/생리학적 작용
PGE1 analog prodrug which is rapidly de-esterified to active "misoprostolic acid". Cited for extremely wide-ranging therapeutic effects, including prevention of NSAID-induced gastric ulceration, regulation of immunologic cascades, inhibition of platelet-activating factor (PAF), treatment of ethanol- and acetaminophen-induced hepatotoxicity and hepatitis, and stimulation of cartilage repair after injury.
포장
The product is delivered as supplied by the issuing Pharmacopoeia. For the current unit quantity, please visit the EDQM reference substance catalogue.
기타 정보
Sales restrictions may apply.
신호어
Danger
유해 및 위험 성명서
예방조치 성명서
Hazard Classifications
Acute Tox. 3 Oral - Repr. 1B
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
가장 최신 버전 중 하나를 선택하세요:
Amanda Selk et al.
Obstetrics and gynecology, 118(4), 941-949 (2011-09-22)
To estimate the benefits and harms of misoprostol use for cervical dilation in patients undergoing operative hysteroscopy. We searched MEDLINE, EMBASE, and the Cochrane Central Register of Controlled Trials (from inception to February 2011). We also searched trial registries, other
C W Ho et al.
Alimentary pharmacology & therapeutics, 37(8), 819-824 (2013-02-26)
Poor adherence to gastroprotective agents (GPAs) is common among users of nonsteroidal anti-inflammatory drugs (NSAIDs) or low-dose aspirin (ASA). There are little data on the utilization of GPAs among NSAID and ASA users complicated by ulcer bleeding. To study the
Deborah Bartz et al.
Obstetrics and gynecology, 122(1), 57-63 (2013-06-08)
To compare the efficacy and acceptability of buccal misoprostol or a synthetic osmotic cervical dilator for cervical preparation before same-day late first-trimester and early second-trimester surgical abortion. In this randomized, double-blind trial, we compared 400 micrograms of buccal misoprostol with
Heleen J van Beekhuizen et al.
International journal of gynaecology and obstetrics: the official organ of the International Federation of Gynaecology and Obstetrics, 122(3), 234-237 (2013-06-25)
To evaluate the efficacy and safety of misoprostol among patients with retained placenta in a low-resource setting. A prospective, multicenter, randomized, double-blind, placebo-controlled trial was carried out in Tanzania between April 2008 and November 2011. It included patients who delivered
N P Polyzos et al.
Human reproduction update, 18(4), 393-404 (2012-05-01)
Although several randomized controlled trials (RCTs) have examined the effect of misoprostol prior to hysteroscopy for cervical dilatation, no solid conclusion has been reached. We therefore set out to perform a meta-analysis of RCTs. We searched MEDLINE, the ISI Web
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.