Y0000703
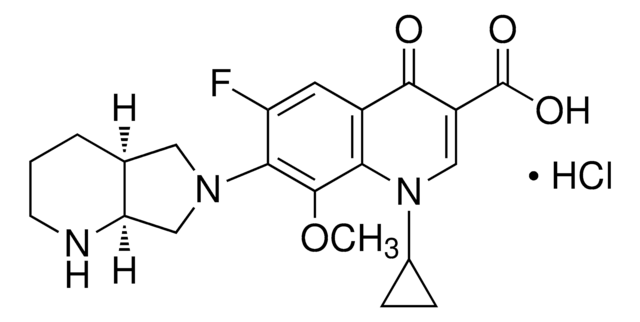
Moxifloxacin hydrochloride
European Pharmacopoeia (EP) Reference Standard
동의어(들):
1-Cyclopropyl-6-fluoro-8-methoxy-7-[(4aS,7aS)-octahydropyrrolo[3,4-b]pyridin-6-yl]-4-oxo-1,4-dihydroquinoline-3-carboxylic acid hydrochloride
About This Item
추천 제품
Grade
pharmaceutical primary standard
API family
moxifloxacin
제조업체/상표
EDQM
응용 분야
pharmaceutical (small molecule)
형식
neat
저장 온도
2-8°C
SMILES string
Cl.COc1c(N2C[C@@H]3CCCN[C@@H]3C2)c(F)cc4C(=O)C(=CN(C5CC5)c14)C(O)=O
InChI
1S/C21H24FN3O4.ClH/c1-29-20-17-13(19(26)14(21(27)28)9-25(17)12-4-5-12)7-15(22)18(20)24-8-11-3-2-6-23-16(11)10-24;/h7,9,11-12,16,23H,2-6,8,10H2,1H3,(H,27,28);1H/t11-,16+;/m0./s1
InChI key
IDIIJJHBXUESQI-DFIJPDEKSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
애플리케이션
포장
기타 정보
관련 제품
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 4 Oral - Aquatic Chronic 3 - Eye Irrit. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
가장 최신 버전 중 하나를 선택하세요:
이미 열람한 고객
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.