PHR1257
Lidocaine hydrochloride
Pharmaceutical Secondary Standard; Certified Reference Material
동의어(들):
Lidocaine hydrochloride monohydrate, 2-Diethylamino-N-(2,6-dimethylphenyl)acetamide hydrochloride monohydrate, Lignocaine hydrochloride monohydrate, Xylocaine hydrochloride monohydrate
About This Item
추천 제품
Grade
certified reference material
pharmaceutical secondary standard
Quality Level
Agency
traceable to BP 214
traceable to Ph. Eur. L0600000
traceable to USP 1366013
API family
lidocaine
CofA
current certificate can be downloaded
기술
HPLC: suitable
gas chromatography (GC): suitable
응용 분야
pharmaceutical (small molecule)
형식
neat
저장 온도
2-30°C
SMILES string
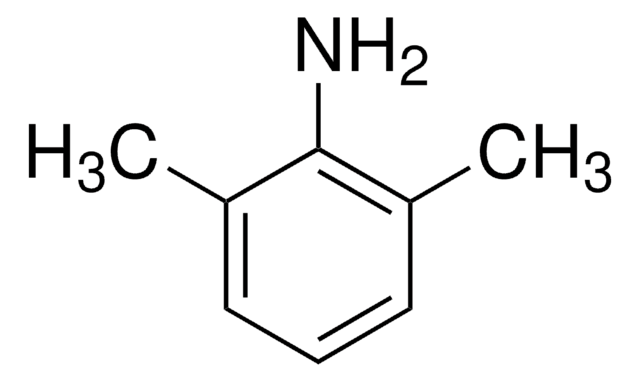
Cl[H].[H]O[H].CCN(CC)CC(=O)Nc1c(C)cccc1C
InChI
1S/C14H22N2O.ClH.H2O/c1-5-16(6-2)10-13(17)15-14-11(3)8-7-9-12(14)4;;/h7-9H,5-6,10H2,1-4H3,(H,15,17);1H;1H2
InChI key
YECIFGHRMFEPJK-UHFFFAOYSA-N
유전자 정보
human ... SCN10A(6336) , SCN11A(11280) , SCN1A(6323) , SCN2A(6326) , SCN3A(6328) , SCN4A(6329) , SCN5A(6331) , SCN7A(6332) , SCN8A(6334) , SCN9A(6335)
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
Pharmaceutical secondary standards for application in quality control, provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards.
Lidocaine hydrochloride is an amide type local anaesthetic that belongs to the class of 1b antiarrhythmics. It is used for regional nerve blocks and infiltrative administration of anaesthesia. It prevents the entry of sodium ions into nerve endings, at the site of pain, and disrupts the electrical signal from reaching the brain.
애플리케이션
- Sensitive determination of lidocaine hydrochloride and cetylpyridinium chloride in their binary mixtures in different pharmaceutical formulations by three spectrophotometric-based methods
- Simultaneous analysis of aminoacridine hydrochloride and lidocaine hydrochloride in bulk powder and pharmaceutical formulation by high-performance liquid chromatography (HPLC) and thin-layer chromatography (TLC)-densitometric methods
- Quantification of ceftriaxone sodium and lidocaine HCl in human plasma samples by high-performance liquid chromatography coupled with tandem mass spectrometry (HPLC-MS/MS)
- Quantitative analysis of lidocaine hydrochloride and miramistin in a wound healing gel sample by high-performance liquid chromatography (HPLC) combined with UV detection
- Development of a method based on liquid–liquid extraction of nifedipine and lidocaine hydrochloride from human plasma samples for their subsequent analysis by high-performance liquid chromatography-tandem mass spectrometry (HPLC-MS/MS)
- Dispersive liquid-liquid microextraction (DLLME) followed by attenuated total reflectance-Fourier transform infrared measurement of dry films for the determination of lidocaine hydrochloride in human urine sample
생화학적/생리학적 작용
분석 메모
각주
추천 제품
관련 제품
신호어
Danger
유해 및 위험 성명서
예방조치 성명서
Hazard Classifications
Acute Tox. 3 Oral
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
Choose from one of the most recent versions:
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.