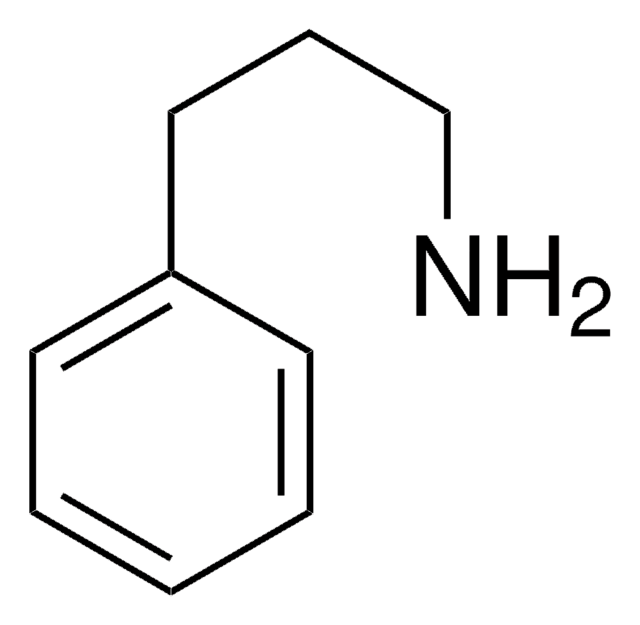
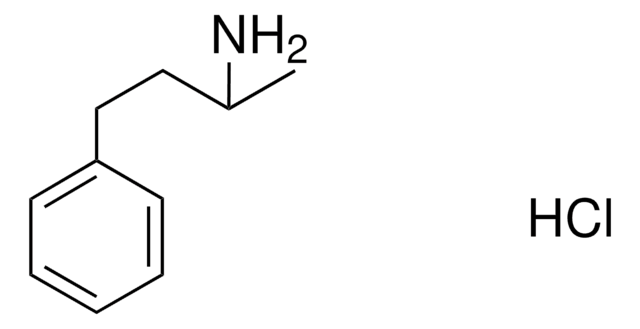
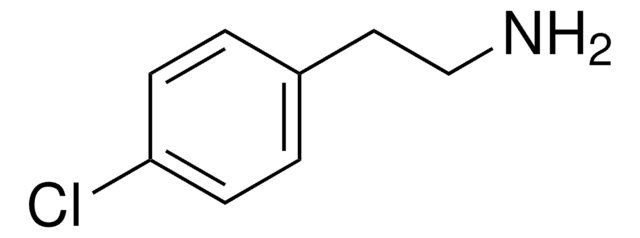
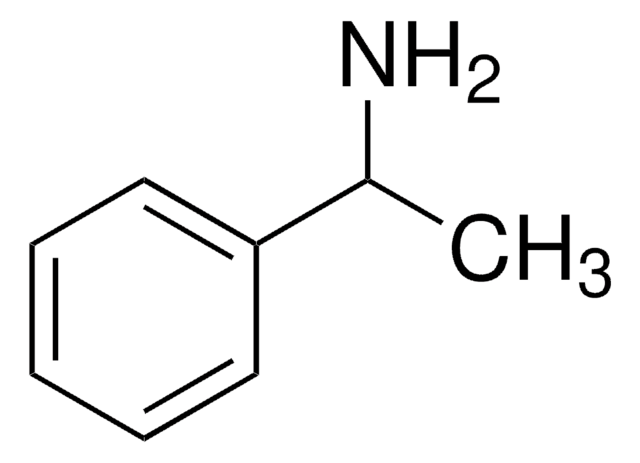
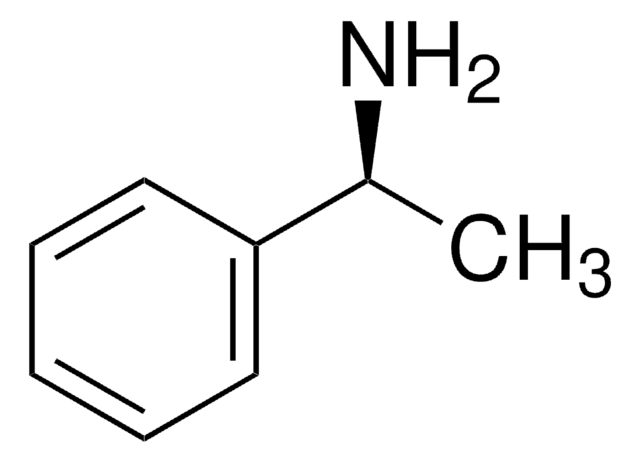
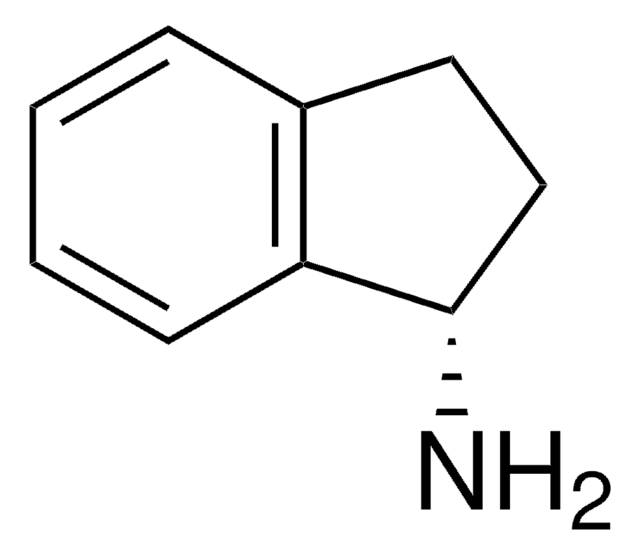
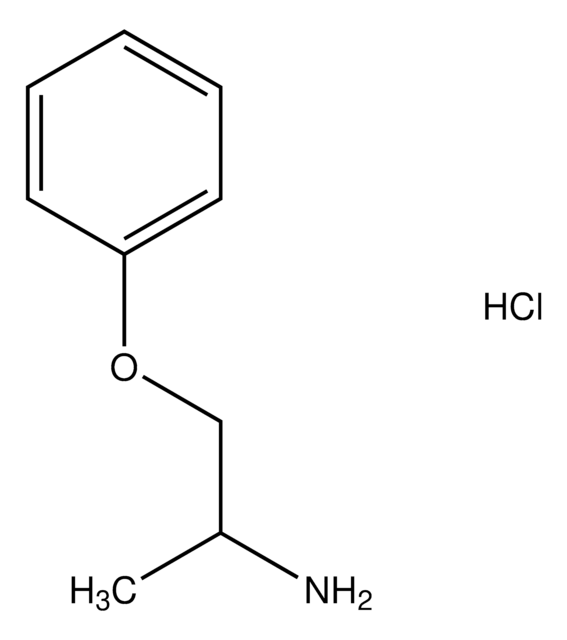
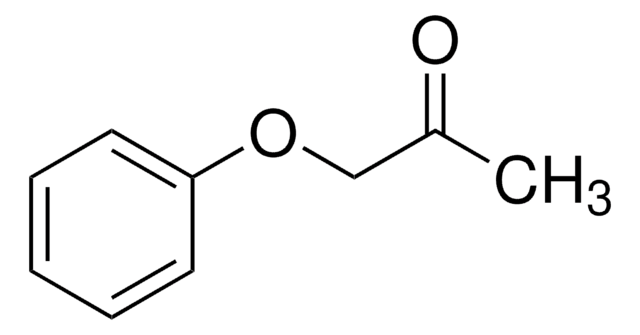
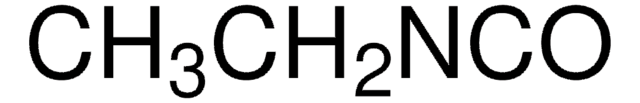추천 제품
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 3 Dermal - Acute Tox. 3 Oral - Aquatic Chronic 3 - Eye Dam. 1 - Skin Corr. 1B
Storage Class Code
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
WGK
WGK 2
Flash Point (°F)
208.0 °F
Flash Point (°C)
97.78 °C
개인 보호 장비
Faceshields, Gloves, Goggles, type ABEK (EN14387) respirator filter
K Yeleswaram et al.
Drug metabolism and disposition: the biological fate of chemicals, 21(2), 284-292 (1993-03-01)
Labetalol causes significant maternal and fetal metabolic effects in pregnant sheep (Yeleswaram et al., J. Pharmacol. Exp. Ther. 262, 683-691 (1992)). This study was undertaken to investigate the contribution of skeletal muscles in the development of metabolic acidosis induced by
Hui Chen et al.
Journal of the American Chemical Society, 141(12), 4963-4971 (2019-03-06)
Enantiomerically pure chiral amines are of increasing value in the preparation of bioactive compounds, pharmaceuticals, and agrochemicals. ω-Transaminase (ω-TA) is an ideal catalyst for asymmetric amination because of its excellent enantioselectivity and wide substrate scope. To shift the equilibrium of
A Changchit et al.
Biological mass spectrometry, 20(12), 751-758 (1991-12-01)
We previously identified 3-amino-1-phenylbutane (APB) as an oxidative N-dealkylated, metabolite of the antihypertensive agent labetalol. Labetalol has two asymmetric centers and is used clinically as a mixture of the four possible stereoisomers; APB has one asymmetric center. We now report
Benedikt Stegmann et al.
Therapeutic drug monitoring, 38(1), 98-107 (2015-09-09)
For psychostimulants, a marked individual variability in the dose-response relationship and large differences in plasma concentrations after similar doses are known. Therefore, optimizing the efficacy of these drugs is at present the most promising way to exploit their full pharmacological
R B Gilbert et al.
Journal of analytical toxicology, 19(2), 84-86 (1995-03-01)
A metabolite of labetalol that is responsible for previous reports of false-positive assays for amphetamines by thin-layer chromatography and immunoassay has been identified. The compound, 3-amino-1-phenylbutane (APB), an oxidative metabolite of labetalol, was initially identified in a patient's urine by
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.