추천 제품
Quality Level
분석
98%
mp
211-213 °C (lit.)
형광
λem 373 nm in methanol
SMILES string
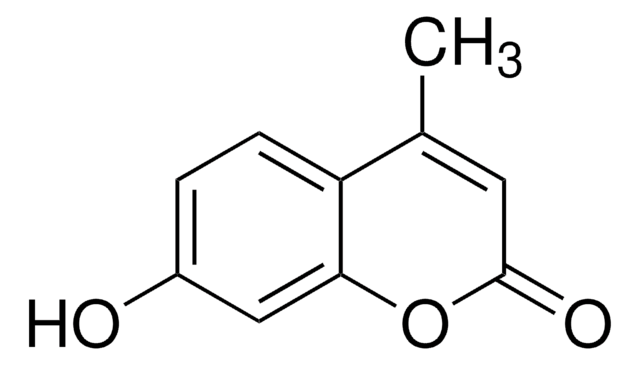
OC1=CC(=O)Oc2ccccc12
InChI
1S/C9H6O3/c10-7-5-9(11)12-8-4-2-1-3-6(7)8/h1-5,10H
InChI key
VXIXUWQIVKSKSA-UHFFFAOYSA-N
유전자 정보
mouse ... Maoa(17161)
rat ... Aldh1a2(116676)
유사한 제품을 찾으십니까? 방문 제품 비교 안내
관련 카테고리
신호어
Warning
유해 및 위험 성명서
예방조치 성명서
Hazard Classifications
Acute Tox. 4 Oral
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
dust mask type N95 (US), Eyeshields, Gloves
이미 열람한 고객
Karen A Nolan et al.
Journal of medicinal chemistry, 52(22), 7142-7156 (2009-11-03)
The synthesis is reported here of two novel series of inhibitors of human NAD(P)H quinone oxidoreductase-1 (NQO1), an enzyme overexpressed in several types of tumor cell. The first series comprises substituted symmetric dicoumarol analogues; the second series contains hybrid compounds
Ayoob Bazgir et al.
Ultrasonics sonochemistry, 17(2), 447-452 (2009-10-20)
A simple, facile, efficient and three-component procedure for the synthesis of spiro[indoline-3,4'-pyrazolo[3,4-b]pyridine]-2,6'(1'H)-diones by the reaction of 4-hydroxycumarin, isatins and 1H-pyrazol-5-amines in water under ultrasonic irradiation is reported. The advantages of this method are the use of an inexpensive and readily
Mikael I Naumov et al.
The Journal of organic chemistry, 72(9), 3293-3301 (2007-03-29)
2-(methoxymethoxymethyl)aryllead triacetates, obtained in situ from the corresponding arylboronic acids, reacted with 4-hydroxycoumarins, leading to 3-(2-methoxymethoxymethyl)aryl-4-hydroxycoumarin derivatives in good to high yields. These compounds underwent a cascade sequence of reactions, deprotection-halogenation-annulation, to afford polyoxygenated tetracyclic 6H,11H-[2]benzopyrano-[4,3-c] [1]benzopyran-11-ones in good yields.
Katrin J Czogalla et al.
Blood, 122(15), 2743-2750 (2013-08-29)
Since the discovery of warfarin-sensitive vitamin K 2,3-epoxide reductase complex subunit 1 (VKORC1), 26 human VKORC1 (hVKORC1) missense mutations have been associated with oral anticoagulant resistance (OACR). Assessment of warfarin resistance using the "classical" dithiothreitol-driven vitamin K 2,3-epoxide reductase (VKOR)
Benye Liu et al.
Plant molecular biology, 72(1-2), 17-25 (2009-09-17)
Coumarin forms in melilotoside (trans-ortho-coumaric acid glucoside)-containing plant species upon cell damage. In moldy melilotoside-containing plant material, trans-ortho-coumaric acid is converted by fungi to 4-hydroxycoumarin, two molecules of which spontaneously combine with formaldehyde to give dicoumarol. Dicoumarol causes internal bleeding
Global Trade Item Number
| SKU | GTIN |
|---|---|
| H23805-100G | 4061833792018 |
| H23805-500G | 4061833553640 |
| H23805-5G |
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.