추천 제품
일반 설명
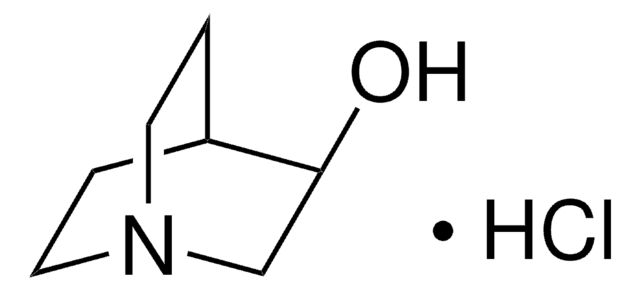
(R)-3-Quinuclidinol is a valuable compound for the production of various pharmaceuticals.
애플리케이션
3-Quinuclidinol has been used:
- as chiral building block for many antimuscarinic agents
- in chemoselective α−iodination of various simple and multi-functionalised acrylic esters via Morita-Baylis-Hillman protocol
- as reagent for cleavage of β-keto and vinylogous β-keto esters
- as synthon for the preparation of cholinergic receptor ligands and anesthetics
- as catalyst for condensation of methyl vinyl ketone with aldehydes
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Skin Corr. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Selective Cleavage of ?-Keto and Vinylogous ?-Keto Esters by 3-Quinuclidinol.
Parish EJ, et al.
Synthetic Communications, 5(5), 341-345 (1975)
Synthesis and local anaesthetic activities of 3-(2-alkoxyphenylcarbamoyloxy)chinuclidinium chlorides.
F Gregán et al.
Die Pharmazie, 48(6), 465-466 (1993-06-01)
Khim.-Farm. Zh., 26, 12-12 (1992)
Tetrahedron Letters, 31, 4509-4509 (1990)
V I Cohen et al.
Journal of medicinal chemistry, 34(10), 2989-2993 (1991-10-01)
Seven analogues of 3-quinuclidinyl benzilate (QNB) in which one phenyl ring was replaced by an alkoxyalkyl moiety were synthesized and their affinities for the muscarinic cholinergic receptor determined. An oxygen in the beta-position of the moiety was not well-tolerated. By
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.

![1,4-Diazabicyclo[2.2.2]octane ReagentPlus®, ≥99%](/deepweb/assets/sigmaaldrich/product/structures/366/129/a6ff4175-974d-4fac-9038-b35e508ef252/640/a6ff4175-974d-4fac-9038-b35e508ef252.png)
