249505
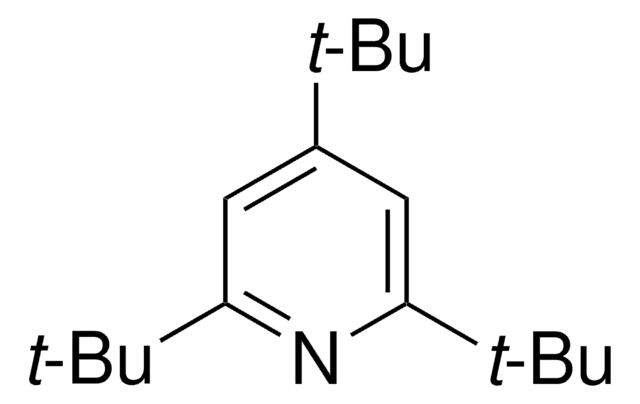
2,6-Di-tert-butyl-4-methylpyridine
98%
동의어(들):
2,6-Bis(1,1-dimethylethyl)-4-methylpyridine, 2,6-Bis(tert-butyl)-4-methylpyridine, 2,6-Ditert-butyl-4-methylpyridine, 4-Methyl-2,6-di-tert-butylpyridine
로그인조직 및 계약 가격 보기
모든 사진(1)
About This Item
실험식(Hill 표기법):
C14H23N
CAS Number:
Molecular Weight:
205.34
Beilstein:
130503
EC Number:
MDL number:
UNSPSC 코드:
12352100
PubChem Substance ID:
NACRES:
NA.22
추천 제품
분석
98%
양식
solid
refractive index
n20/D 1.4763 (lit.)
bp
233 °C (lit.)
mp
33-36 °C (lit.)
solubility
ethanol: soluble 5%, clear to slightly hazy, colorless to dark yellow
저장 온도
2-8°C
SMILES string
Cc1cc(nc(c1)C(C)(C)C)C(C)(C)C
InChI
1S/C14H23N/c1-10-8-11(13(2,3)4)15-12(9-10)14(5,6)7/h8-9H,1-7H3
InChI key
HVHZEKKZMFRULH-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
2,6-Di-tert-butyl-4-methylpyridine is a sterically hindered, non-nucleophilic base which distinguishes between BrÖnsted (protonic) and Lewis acids. It also enables the direct high-yield conversion of aldehydes and ketones to vinyl triflates. It inhibits desilylation and hydration of the products during GaCl3-catalyzed ortho-ethynylation of phenol.
애플리케이션
2,6-Di-tert-butyl-4-methylpyridine has been used:
- in the synthesis of 1,2-dihydro-2-silanaphthalene derivatives
- as base in PtCl4-catalyzed cyclization reactions of homopropargyl azide derivatives
- diastereoselective synthesis of β-thiomannopyranosides
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
표적 기관
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
183.2 °F - closed cup
Flash Point (°C)
84 °C - closed cup
개인 보호 장비
dust mask type N95 (US), Eyeshields, Gloves
이미 열람한 고객
Hidekazu Arii et al.
Chemical communications (Cambridge, England), 50(50), 6649-6652 (2014-05-16)
Treatment of dialkylbenzylsilane (1) with trityl tetrakis(pentafluorophenyl)borate (TPFPB) in the presence of terminal or internal alkynes (3) and 2,6-di-tert-butyl-4-methylpyridine gave the corresponding 1,2-dihydro-2-silanaphthalene derivatives (4) in 34-82% yields.
Organic Syntheses, 68, 138-138 (1990)
Synthesis, 283-283 (1980)
Daichi Nakamura et al.
Organic & biomolecular chemistry, 17(14), 3581-3589 (2019-03-23)
Regioselectivity of Ln(OTf)3-catalysed alcoholysis of 2,3- and 3,4-epoxy alcohols was closely investigated to expand the scope of the transformations. The synthetic use was demonstrated by application to the construction of 4-propoxy-5-hydroxy-2,3-pentanedione (C4-propoxy-HPD), which is a potent synthetic mediator in AI-2
D Crich et al.
The Journal of organic chemistry, 65(3), 801-805 (2000-05-18)
A highly diastereoselective synthesis of beta-thiomannopyranosides is described in which S-phenyl 2,3-di-O-benzyl-4, 6-O-benzylidene-1-deoxy-1-thia-alpha-D-mannopyranoside S-oxide is treated with triflic anhydride and 2, 6-di-tert-butyl-4-methylpyridine in CH(2)Cl(2) at -78 degrees C leading to the formation of an intermediate alpha-mannosyl triflate. Addition of primary
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.