246948
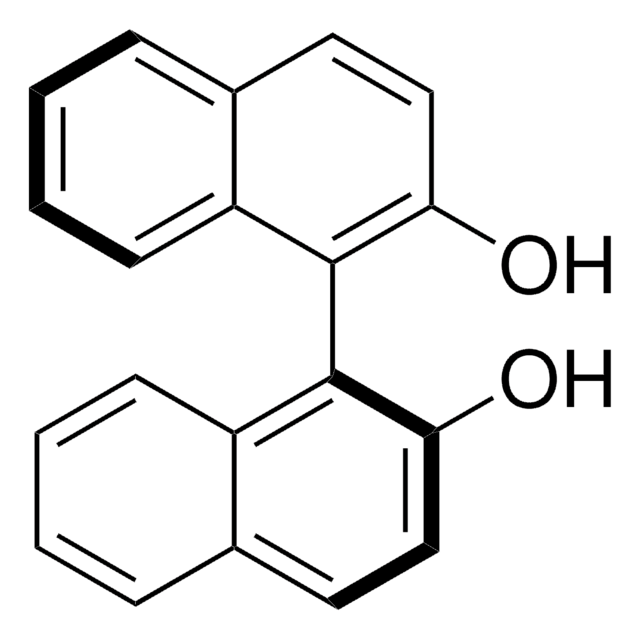
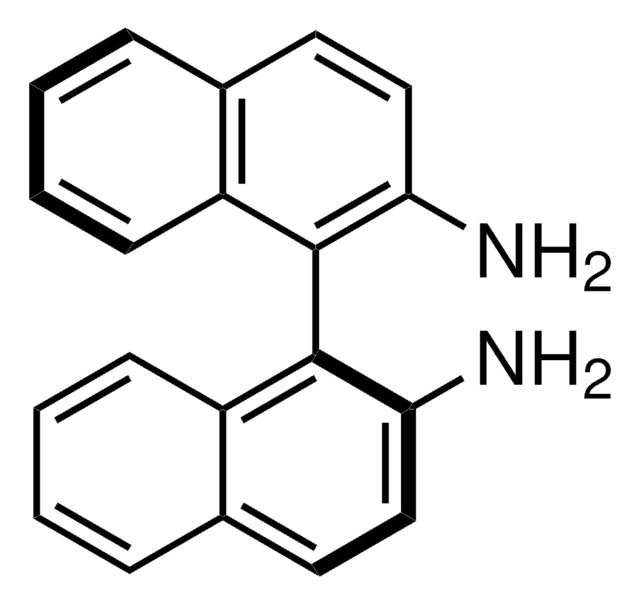
(R)-(+)-1,1′-Bi(2-naphthol)
99%
동의어(들):
(+)-2,2′-Dihydroxy-1,1′-dinaphthyl, (R)-(+)-1,1′-Binaphthalene-2,2′-diol, (R)-BINOL
로그인조직 및 계약 가격 보기
모든 사진(3)
About This Item
Linear Formula:
HOC10H6C10H6OH
CAS Number:
Molecular Weight:
286.32
Beilstein:
3616837
MDL number:
UNSPSC 코드:
12352104
PubChem Substance ID:
EC 인덱스 번호:
606-048-4
NACRES:
NA.22
추천 제품
분석
99%
형태
solid
광학 활성
[α]21/D +34°, c = 1 in THF
광학 순도
ee: 99% (HPLC)
mp
208-210 °C (lit.)
SMILES string
Oc1ccc2ccccc2c1-c3c(O)ccc4ccccc34
InChI
1S/C20H14O2/c21-17-11-9-13-5-1-3-7-15(13)19(17)20-16-8-4-2-6-14(16)10-12-18(20)22/h1-12,21-22H
InChI key
PPTXVXKCQZKFBN-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
애플리케이션
A chiral auxiliary used in the catalytic asymmetric oxidation of sulfides to sulfoxides. Chiral lanthanide triflates formed from binaphthol serve as catalysts for asymmetric Diels-Alder reactions. Derivatives of binaphthol have recently found use in asymmetric Claisen rearrangements and asymmetric epoxidations. The lithium aluminum hydride derivative of these diols (BINAP-H) has been used extensively for the reduction of ketones.
Chiral binapthol imminium salt precursor. Salts were used for an asymmetric epoxidation of olefins.
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Faceshields, Gloves, type P2 (EN 143) respirator cartridges
이미 열람한 고객
Asymmetric aza-Diels-Alder reaction catalyzed by boron reagent: Effect of biphenol and binaphthol ligand.
Hattori, K. Yamamoto, H.
Synlett, 1993(02), 129-129 (1993)
Asymmetric Diels-Alder reactions catalyzed by chiral lanthanide (III) trifluoromethanesulfonates. Unique structure of the triflate and stereoselective synthesis of both enantiomers using a single chiral source and a choice of achiral ligands.
S. Kobayashi et al.
Tetrahedron, 50(40), 11623-11623 (1994)
Catalytic asymmetric oxidation of sulfides to sulfoxides with tert-butyl hydroperoxide using binaphthol as a chiral auxiliary.
Komatsu N, et al.
The Journal of Organic Chemistry, 58, 4529-4529 (1993)
Catalytic asymmetric epoxidation with (Salen) manganese (III) complex bearing binaphthyl groups of axial chirality.
Sasaki, H. et al.
Synlett, 1993(04), 300-300 (1993)
New chiral binaphthalene-derived iminium salt organocatalysts for asymmetric epoxidation of alkenes.
Philip C Bulman Page et al.
The Journal of organic chemistry, 72(12), 4424-4430 (2007-05-18)
A series of binaphthalene-fused azepinium salts has been generated and used as organocatalysts in the asymmetric epoxidation of unfunctionalized alkenes, giving rise to ees of up to 84%.
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.