おすすめの製品
グレード
pharmaceutical primary standard
APIファミリー
nifedipine
メーカー/製品名
USP
アプリケーション
pharmaceutical (small molecule)
フォーマット
neat
SMILES記法
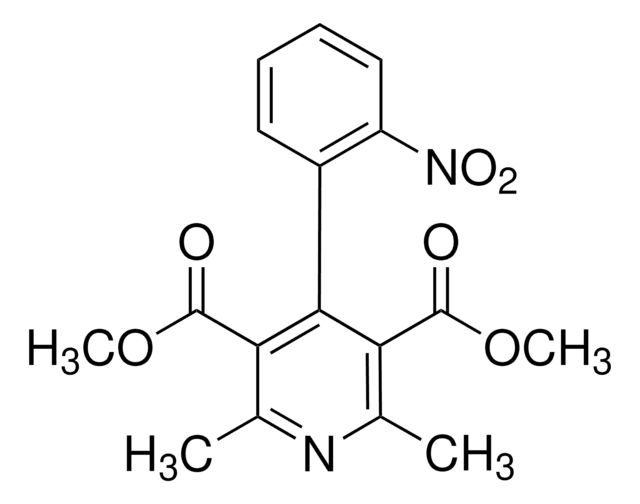
COC(=O)c1c(C)nc(C)c(C(=O)OC)c1-c2ccccc2[N+]([O-])=O
InChI
1S/C17H16N2O6/c1-9-13(16(20)24-3)15(14(10(2)18-9)17(21)25-4)11-7-5-6-8-12(11)19(22)23/h5-8H,1-4H3
InChI Key
UMQHJQGNGLQJPF-UHFFFAOYSA-N
類似した製品をお探しですか? 訪問 製品比較ガイド
アプリケーション
Nifedipine Nitrophenylpyridine Analog USP Reference standard, intended for use in specified quality tests and assays as specified in the USP compendia. Also, for use with USP monographs such as:
- Nifedipine Capsules
- Nifedipine
- Nifedipine Extended-Release Tablets
生物化学的/生理学的作用
CYP3A4によるニフェジピンの代謝物です。親化合物であるニフェジピンは、抗狭心症薬および血圧降下薬です。
アナリシスノート
These products are for test and assay use only. They are not meant for administration to humans or animals and cannot be used to diagnose, treat, or cure diseases of any kind.
その他情報
Sales restrictions may apply.
関連製品
製品番号
詳細
価格
シグナルワード
Danger
危険有害性情報
危険有害性の分類
Eye Dam. 1
保管分類コード
11 - Combustible Solids
WGK
WGK 2
引火点(°F)
Not applicable
引火点(℃)
Not applicable
適用法令
試験研究用途を考慮した関連法令を主に挙げております。化学物質以外については、一部の情報のみ提供しています。 製品を安全かつ合法的に使用することは、使用者の義務です。最新情報により修正される場合があります。WEBの反映には時間を要することがあるため、適宜SDSをご参照ください。
Jan Code
1463600-25MG:
最新バージョンのいずれかを選択してください:
Wenzhan Yang et al.
Current drug discovery technologies, 5(2), 129-139 (2008-08-05)
This study reports the use of para-sulphonato calix[8]arene to produce stable complexes with improved bioavailability for nifedipine, a calcium-channel blocker that is practically insoluble in water. Thermal analysis and electrospray ionisation mass spectroscopy confirmed that nifedipine formed complexes with the
Tsai-Shin Chiang et al.
PloS one, 9(4), e94885-e94885 (2014-04-16)
Human hepatoma cell lines are commonly used as alternatives to primary hepatocytes for the study of drug metabolism in vitro. However, the phase I cytochrome P450 (CYP) enzyme activities in these cell lines occur at a much lower level than
Camille C Savary et al.
Drug metabolism and disposition: the biological fate of chemicals, 42(8), 1235-1240 (2014-05-17)
Humans are usually exposed to several pesticides simultaneously; consequently, combined actions between pesticides themselves or between pesticides and other chemicals need to be addressed in the risk assessment. Many pesticides are efficient activators of pregnane X receptor (PXR) and/or constitutive
Yohei Kosugi et al.
Xenobiotica; the fate of foreign compounds in biological systems, 45(4), 345-352 (2014-11-12)
1. The purpose of this study was to clarify species differences in the heteroactivation of CYP3A substrates by efavirenz, which is known from clinical studies to activate midazolam 1'-hydroxylation, and to assess the feasibility of an animal model. 2. In monkey and
Yoshihiko Shimokawa et al.
Biological & pharmaceutical bulletin, 37(11), 1727-1735 (2014-11-05)
Delamanid is a new drug for the treatment of multidrug-resistant tuberculosis. Individuals who are co-infected with human immunodeficiency virus and Mycobacterium tuberculosis may require treatment with a number of medications that might interact significantly with the CYP enzyme system as
ライフサイエンス、有機合成、材料科学、クロマトグラフィー、分析など、あらゆる分野の研究に経験のあるメンバーがおります。.
製品に関するお問い合わせはこちら(テクニカルサービス)