おすすめの製品
由来生物
synthetic (organic)
アッセイ
≥98%
フォーム
powder
不純物
inosine, essentially free
溶解性
1 M NH4OH: 50 mg/mL, clear, colorless
保管温度
−20°C
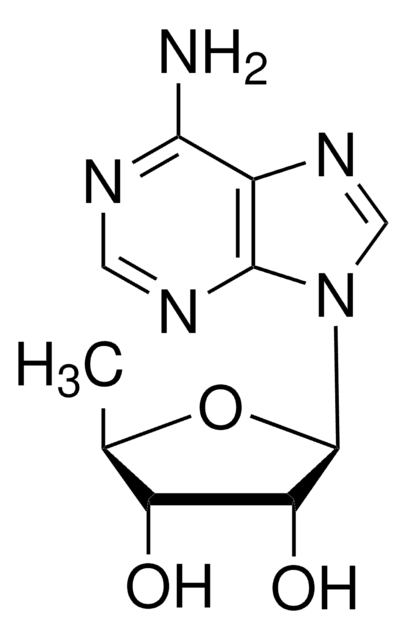
SMILES記法
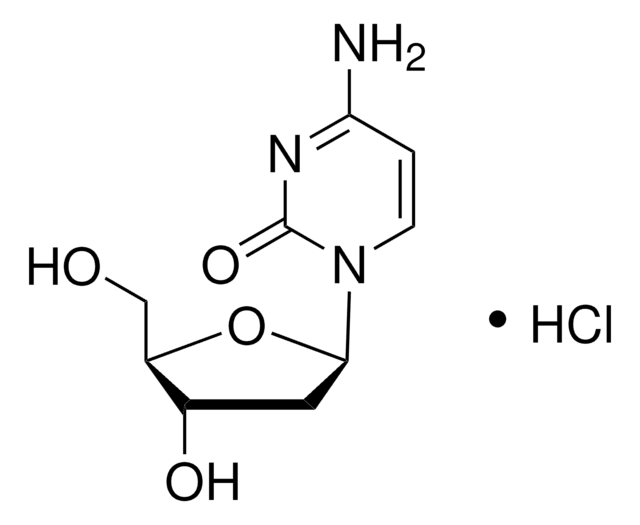
OC[C@H]1O[C@H](C[C@@H]1O)n2cnc3C(=O)NC=Nc23
InChI
1S/C10H12N4O4/c15-2-6-5(16)1-7(18-6)14-4-13-8-9(14)11-3-12-10(8)17/h3-7,15-16H,1-2H2,(H,11,12,17)/t5-,6+,7+/m0/s1
InChI Key
VGONTNSXDCQUGY-RRKCRQDMSA-N
類似した製品をお探しですか? 訪問 製品比較ガイド
アプリケーション
2′-Deoxyinosine has been used in the quantification of nucleoside forms of DNA lesion in a single DNA sample by liquid chromatography tandem mass spectrometry (LC-MS/MS). It has also been used as a standard for high performance liquid chromatography analysis.
生物化学的/生理学的作用
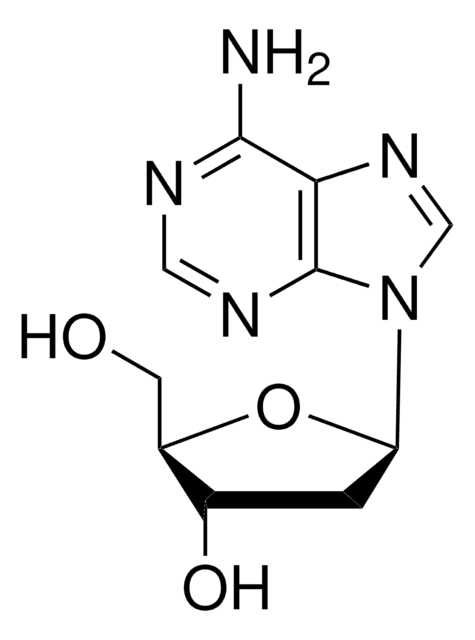
2′-Deoxyinosine is a nucleoside composed of hypoxanthine attached to 2′-deoxyribose via a β-N9-glycosidic bond. 2′-Deoxyinosine in DNA can arise from deamination of adenosine. 2′-deoxyinsine can be used as a model compound to study the chemistry of adduct formation and radical chemistry that may affect DNA structures. 2′-Deoxyinosine is used to produce hybridization-sensitive fluorescent DNA probes with self-avoidance ability.
2′-Deoxyinosine is a nucleoside form of hypoxanthine. It is a DNA damage product resulting from the impairment of DNA by reactive nitrogen species. 2′-deoxyinosine is formed from nitrosative deamination by N2O3.
保管分類コード
11 - Combustible Solids
WGK
WGK 3
引火点(°F)
Not applicable
引火点(℃)
Not applicable
個人用保護具 (PPE)
Eyeshields, Gloves, type N95 (US)
適用法令
試験研究用途を考慮した関連法令を主に挙げております。化学物質以外については、一部の情報のみ提供しています。 製品を安全かつ合法的に使用することは、使用者の義務です。最新情報により修正される場合があります。WEBの反映には時間を要することがあるため、適宜SDSをご参照ください。
Jan Code
D5287-VAR:
D5287-5G:
D5287-BULK:
D5287-1G:
D5287-500MG:
D5287-250MG:
D5287-100MG:
この製品を見ている人はこちらもチェック
Shuji Ikeda et al.
Organic & biomolecular chemistry, 8(3), 546-551 (2010-01-22)
Hybridization-sensitive fluorescent probes have an inherent disadvantage: self-dimerization of the probe prevents the fluorescence quenching prior to hybridization with the target, resulting in a high background signal. To avoid self-dimerization of probes, we focused on a base pair formed by
Quantification of DNA damage products resulting from deamination, oxidation and reaction with products of lipid peroxidation by liquid chromatography isotope dilution tandem mass spectrometry
Taghizadeh K, et al.
Nature Protocols, 3(8), 1287-1287 (2008)
Lipid peroxidation dominates the chemistry of DNA adduct formation in a mouse model of inflammation.
Bo Pang et al.
Carcinogenesis, 28(8), 1807-1813 (2007-03-10)
In an effort to define the prevalent DNA damage chemistry-associated chronic inflammation, we have quantified 12 DNA damage products in tissues from the SJL mouse model of nitric oxide (NO) overproduction. Using liquid chromatography-mass spectrometry/MS and immunoblot techniques, we analyzed
Rongjuan Mi et al.
Mutation research, 735(1-2), 12-18 (2012-06-06)
The human endonuclease V gene is located in chromosome 17q25.3 and encodes a 282 amino acid protein that shares about 30% sequence identity with bacterial endonuclease V. This study reports biochemical properties of human endonuclease V with respect to repair
A Cohen et al.
The New England journal of medicine, 295(26), 1449-1454 (1976-12-23)
To delineate the normal function of purine nucleoside phosphorylase and to understand the pathogenesis of the immune dysfunction associated with deficiency of this enzyme, we studied purine metabolism in a patient deficient in purine nucleoside phosphorylase, her erythrocytes and cultured
ライフサイエンス、有機合成、材料科学、クロマトグラフィー、分析など、あらゆる分野の研究に経験のあるメンバーがおります。.
製品に関するお問い合わせはこちら(テクニカルサービス)