Y0001090
フルバスタチン ナトリウム
European Pharmacopoeia (EP) Reference Standard
別名:
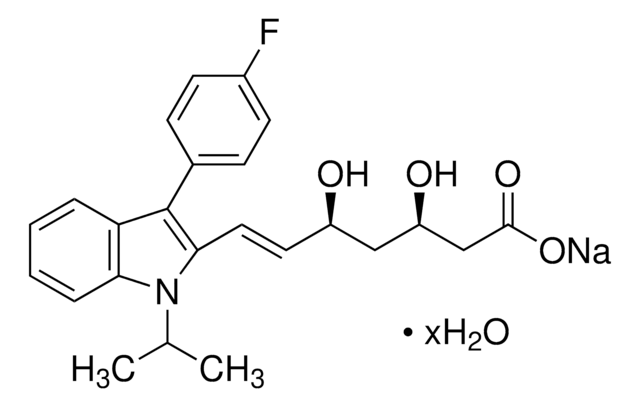
(3R,5S,6E)-rel-7-[3-(4-Fluorophenyl)-1-(1-methylethyl)-1H-indol-2-yl]-3,5-dihydroxy-6-heptenoic acid monosodium salt
ログイン組織・契約価格を表示する
すべての画像(1)
About This Item
実験式(ヒル表記法):
C24H25FNNaO4
CAS番号:
分子量:
433.45
MDL番号:
UNSPSCコード:
41116107
PubChem Substance ID:
NACRES:
NA.24
おすすめの製品
グレード
pharmaceutical primary standard
APIファミリー
fluvastatin
メーカー/製品名
EDQM
アプリケーション
pharmaceutical (small molecule)
フォーマット
neat
保管温度
2-8°C
SMILES記法
[Na+].CC(C)n1c(\C=C\[C@H](O)C[C@H](O)CC([O-])=O)c(-c2ccc(F)cc2)c3ccccc13
InChI
1S/C24H26FNO4.Na/c1-15(2)26-21-6-4-3-5-20(21)24(16-7-9-17(25)10-8-16)22(26)12-11-18(27)13-19(28)14-23(29)30;/h3-12,15,18-19,27-28H,13-14H2,1-2H3,(H,29,30);/q;+1/p-1/b12-11+;/t18-,19-;/m0./s1
InChI Key
ZGGHKIMDNBDHJB-RPQBTBOMSA-M
類似した製品をお探しですか? 訪問 製品比較ガイド
詳細
この製品は薬局方標準品です。発行元の薬局方により製造・供給されています。MSDSを含む製品情報などの詳しい情報は、発行元の薬局方のウェブサイトよりご確認ください。
アプリケーション
Fluvastatin sodium EP Reference standard, intended for use in laboratory tests only as specifically prescribed in the European Pharmacopoeia.
包装
The product is delivered as supplied by the issuing Pharmacopoeia. For the current unit quantity, please visit the EDQM reference substance catalogue.
その他情報
Sales restrictions may apply.
関連製品
製品番号
詳細
価格
保管分類コード
11 - Combustible Solids
WGK
WGK 3
引火点(°F)
Not applicable
引火点(℃)
Not applicable
最新バージョンのいずれかを選択してください:
Ramakrishna Nirogi et al.
Journal of chromatography. B, Analytical technologies in the biomedical and life sciences, 907, 65-73 (2012-10-06)
Deferasirox is an iron chelating agent for the treatment of transfusional iron over load in patients with chronic anemia. These anemic patients require close monitoring of the deferasirox exposures for ensuring its therapeutic efficacy. Dried blood spot (DBS) sampling methodology
H R Xu et al.
Journal of biomedicine & biotechnology, 2012, 386230-386230 (2012-07-20)
The aim of this study was to evaluate the difference in pharmacokinetics and pharmacodynamics between extended-release (ER) fluvastatin tablet and its immediate-release (IR) capsule in Chinese healthy subjects. This was an open-label, single/multiple-dose, two-period, two-treatment, crossover, randomized trial with a
Masanori Atsukawa et al.
Journal of gastroenterology and hepatology, 28(1), 51-56 (2012-09-20)
Although the anti-hepatitis C virus (HCV) effect of statins in vitro and clinical efficacy of fluvastatin combined with Pegylated interferon (PEG-IFN)/ribavirin therapy for chronic hepatitis C (CHC) have been reported, the details of clinical presentation are largely unknown. We focused
C Kondo et al.
Journal of viral hepatitis, 19(9), 615-622 (2012-08-07)
Pegylated interferon (PEG-IFN)/ribavirin combination therapy is the standard-of-care (SOC) treatment for chronic hepatitis C patients infected with hepatitis C virus (HCV) genotype 1b and high viral load. The addition of fluvastatin to SOC treatment has been suggested to be effective
Allen B Williams et al.
Blood, 120(15), 3069-3079 (2012-08-29)
FLT3 is frequently mutated in acute myeloid leukemia (AML), but resistance has limited the benefit of tyrosine kinase inhibitors (TKI). We demonstrate that statins can impair FLT3 glycosylation, thus leading to loss of surface expression and induction of cell death
ライフサイエンス、有機合成、材料科学、クロマトグラフィー、分析など、あらゆる分野の研究に経験のあるメンバーがおります。.
製品に関するお問い合わせはこちら(テクニカルサービス)