すべての画像(1)
About This Item
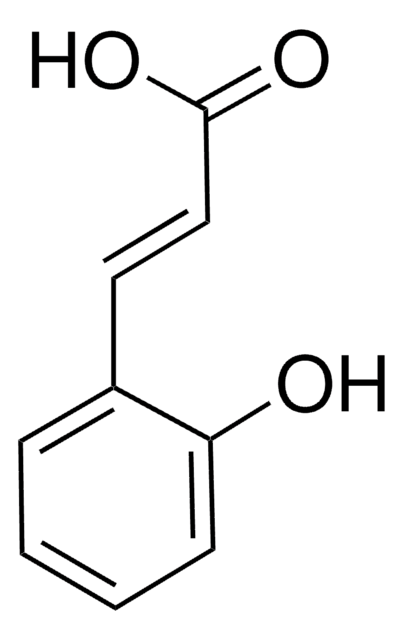
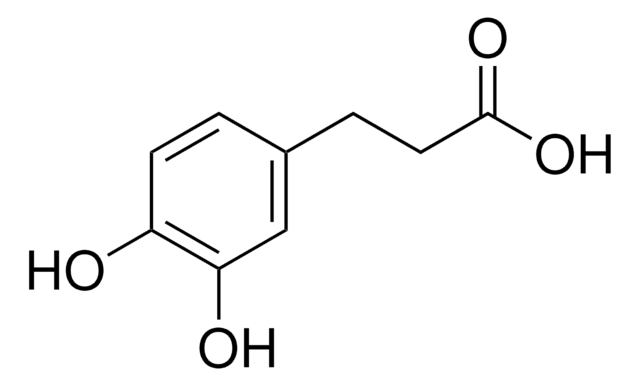
化学式:
HOC6H4CH2CH2CO2H
CAS番号:
分子量:
166.17
MDL番号:
UNSPSCコード:
12352100
PubChem Substance ID:
NACRES:
NA.22
おすすめの製品
アッセイ
99%
mp
86-89 °C (lit.)
官能基
carboxylic acid
SMILES記法
OC(=O)CCc1ccccc1O
InChI
1S/C9H10O3/c10-8-4-2-1-3-7(8)5-6-9(11)12/h1-4,10H,5-6H2,(H,11,12)
InChI Key
CJBDUOMQLFKVQC-UHFFFAOYSA-N
関連するカテゴリー
詳細
3-(2-Hydroxyphenyl) propionic acid is a phenyl propionic acid derivative. It was found to be one of the constituents of Justicia pectoralis Jacq. extract which was analyzed by gas chromatography/mass spectrometry (GC/MS). The antiulcerogenic effect of 3-(2-hydroxyphenyl) propionic acid in the prevention of serotonin-induced ulcerogenesis has been studied in rats. It has been reported to be one of the major microbial metabolite of both (+)-catechin and (-)-epicatechin by human faecal microbiota. Crystal structure study reveals that 3-(2-hydroxyphenyl)propionic acid crystals are monoclinic with space group P21/c.
アプリケーション
3-(2-Hydroxyphenyl)propionic acid is suitable as a growth substrate for various strains of E. coli and as a standard in the study of microbial metabolism of catechin stereoisomers.
シグナルワード
Warning
危険有害性情報
危険有害性の分類
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
ターゲットの組織
Respiratory system
保管分類コード
11 - Combustible Solids
WGK
WGK 3
個人用保護具 (PPE)
dust mask type N95 (US), Eyeshields, Gloves
適用法令
試験研究用途を考慮した関連法令を主に挙げております。化学物質以外については、一部の情報のみ提供しています。 製品を安全かつ合法的に使用することは、使用者の義務です。最新情報により修正される場合があります。WEBの反映には時間を要することがあるため、適宜SDSをご参照ください。
Jan Code
393533-50G:
393533-BULK:
393533-VAR:
393533-10G:
Microbial metabolism of catechin stereoisomers by human faecal microbiota: comparison of targeted analysis and a non-targeted metabolomics method.
Aura AM, et al.
Phytochemistry Letters, 1(1), 18-22 (2008)
R Burlingame et al.
Journal of bacteriology, 155(1), 113-121 (1983-07-01)
A number of laboratory strains and clinical isolates of Escherichia coli utilized several aromatic acids as sole sources of carbon for growth. E. coli K-12 used separate reactions to convert 3-phenylpropionic and 3-(3-hydroxyphenyl)propionic acids into 3-(2,3-dihydroxyphenyl)propionic acid which, after meta-fission
S Tanaka et al.
Planta medica, 55(3), 245-248 (1989-06-01)
Two active compounds that prevent serotonin-induced ulcerogenesis in rats were isolated from Chinese cinnamon (the stem bark of Cinnamomum cassia) and identified as 3-(2-hydroxyphenyl)-propanoic acid and its O-glucoside. The former compound, administered orally or parenterally to rats at a remarkably
J X de Vries et al.
Biomedical & environmental mass spectrometry, 15(8), 413-417 (1988-04-15)
The analysis of extracts from the South American plant Justicia pectoralis Jacq. permitted the identification, among other compounds, of coumarin, dihydrocoumarin, umbelliferone and 3-(2-hydroxyphenyl)propionic acid by gas chromatography/mass spectrometry (GC/MS); the acids and phenolic compounds were derivatized with diazomethane. GC/MS
Structure of 3-(2-hydroxyphenyl) propionic acid.
Begum NS, et al.
Acta Crystallographica Section C, Crystal Structure Communications, 48(6), 1076-1078 (1992)
ライフサイエンス、有機合成、材料科学、クロマトグラフィー、分析など、あらゆる分野の研究に経験のあるメンバーがおります。.
製品に関するお問い合わせはこちら(テクニカルサービス)