70221
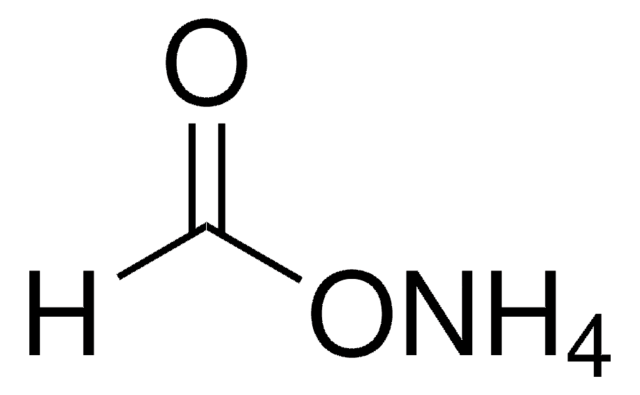
Ammonium formate
eluent additive for LC-MS, LiChropur™, ≥99.0%
Synonym(s):
Formic acid ammonium salt
About This Item
Recommended Products
grade
eluent additive for LC-MS
Quality Level
description
anionic
Assay
≥99.0% (calc. on dry substance, NT)
≥99.0%
form
crystals
quality
LiChropur™
technique(s)
LC/MS: suitable
impurities
insoluble matter, passes filter test
<2% water
ign. residue (900 °C)
<0.1% (as SO4)
pH
5.5-7.5 (25 °C, 1 M in H2O)
mp
119-121 °C (lit.)
solubility
H2O: 1 M at 20 °C, clear, colorless
density
1.26 g/mL at 25 °C (lit.)
anion traces
chloride (Cl-): ≤50 mg/kg
sulfate (SO42-): ≤50 mg/kg
cation traces
Al: ≤1 mg/kg
As: ≤0.1 mg/kg
Ba: ≤1 mg/kg
Bi: ≤1 mg/kg
Ca: ≤5 mg/kg
Cd: ≤1 mg/kg
Co: ≤1 mg/kg
Cr: ≤1 mg/kg
Cu: ≤1 mg/kg
Fe: ≤1 mg/kg
K: ≤5 mg/kg
Li: ≤1 mg/kg
Mg: ≤1 mg/kg
Mn: ≤1 mg/kg
Mo: ≤1 mg/kg
Na: ≤5 mg/kg
Ni: ≤1 mg/kg
Pb: ≤1 mg/kg
Sr: ≤1 mg/kg
Zn: ≤1 mg/kg
λ
1 M in H2O
UV absorption
λ: 260 nm Amax: ≤0.01
λ: 280 nm Amax: ≤0.01
SMILES string
N.OC=O
InChI
1S/CH2O2.H3N/c2-1-3;/h1H,(H,2,3);1H3
InChI key
VZTDIZULWFCMLS-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
Ammonium formate has been used in the separation of non-steroidal anti-inflammatory drugs and of oligosaccharide mixtures using capillary electrochromatography. It is also used in the analysis of phosphatidylserines, triacylglycerols and triacylglycerol oxidation products, and oligogalacturonic acids using HPLC and HPLC-MS methods.
Legal Information
Application
related product
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 1
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Regulatory Listings
Regulatory Listings are mainly provided for chemical products. Only limited information can be provided here for non-chemical products. No entry means none of the components are listed. It is the user’s obligation to ensure the safe and legal use of the product.
JAN Code
70221-100G-F:
70221-BULK-F:
70221-VAR-F:
70221-25G-F:
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Review of clinically relevant ethanol metabolites and analytical method development for the analysis of ethyl glucuronide and ethyl sulfate in urine matrix.
Separation of null
Separation of Oxazepam; Desmethyldiazepam, analytical standard; Temazepam, analytical standard; Diazepam
Protocols
HPLC Analysis of PEG on Two Zenix® SEC-300 Columns in Tandem
Separation of null
Separation of null
Separation of Sorbitol; Mannitol
Chromatograms
application for HPLCapplication for HPLCapplication for HPLCapplication for HPLCShow MoreOur team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service