N9284
Nitroreductase from Escherichia coli
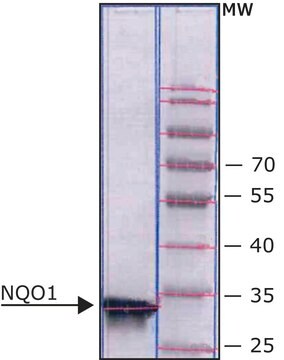
≥90% (SDS-PAGE), recombinant, expressed in E. coli
Sinonimo/i:
NTRA
About This Item
Prodotti consigliati
Ricombinante
expressed in E. coli
Livello qualitativo
Saggio
≥90% (SDS-PAGE)
Forma fisica
lyophilized powder
Attività specifica
≥100 units/mL
PM
monomer 24000
Caratteristiche più verdi
Waste Prevention
Safer Solvents and Auxiliaries
Design for Energy Efficiency
Learn more about the Principles of Green Chemistry.
sustainability
Greener Alternative Product
N° accesso UniProt
Categoria alternativa più verde
Condizioni di spedizione
wet ice
Temperatura di conservazione
−20°C
Informazioni sul gene
Escherichia coli K12 ... nfsB(945483)
Descrizione generale
Applicazioni
Azioni biochim/fisiol
Proprietà fisiche
Definizione di unità
Nota sulla preparazione
Codice della classe di stoccaggio
13 - Non Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Certificati d'analisi (COA)
Cerca il Certificati d'analisi (COA) digitando il numero di lotto/batch corrispondente. I numeri di lotto o di batch sono stampati sull'etichetta dei prodotti dopo la parola ‘Lotto’ o ‘Batch’.
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.![Nitrate Reductase (NAD[P]H) from Aspergillus niger lyophilized powder, ≥300 units/g solid](/deepweb/assets/sigmaaldrich/product/images/309/282/2a67ae4d-ca55-4f0b-96ec-34748ff8a21e/640/2a67ae4d-ca55-4f0b-96ec-34748ff8a21e.jpg)