C2389
Cerulenin
from Cephalosporium caerulens, ≥98% (HPLC), powder, de novo phospholipid synthesis inhibitor
Sinonimo/i:
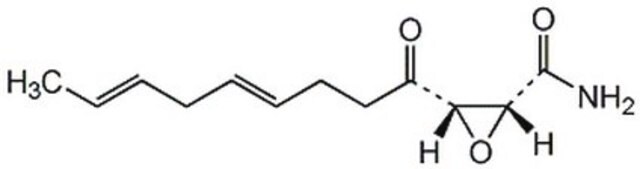
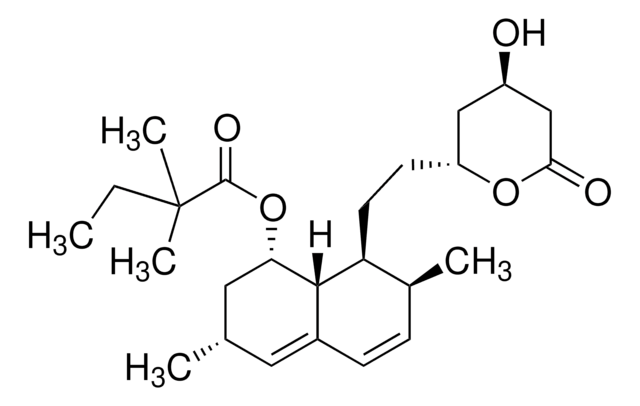
Helicocerin, (2R,3S,E,E)-2,3-Epoxy-4-oxo-7,10-dodecadienamide
About This Item
Prodotti consigliati
Nome del prodotto
Cerulenin, ≥98% (HPLC), from Cephalosporium caerulens
Origine biologica
Cephalosporium caerulens
Livello qualitativo
Saggio
≥98% (HPLC)
Stato
powder
Punto di fusione
93 °C
Solubilità
acetone: 19.60-20.40 mg/mL, clear to slightly hazy, colorless to yellow
Spettro attività antibiotica
fungi
Modalità d’azione
enzyme | inhibits
Temperatura di conservazione
−20°C
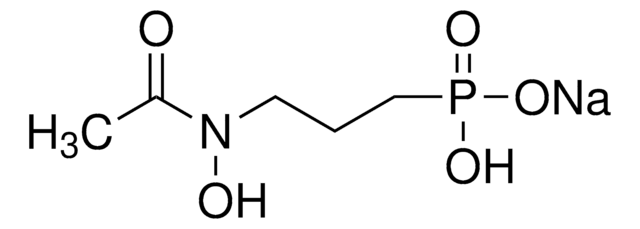
Stringa SMILE
C/C=C/C/C=C/CCC([C@@H]1[C@H](C(N)=O)O1)=O
InChI
1S/C13H18O3/c1-3-4-5-6-7-8-9-11(15)13-12(16-13)10(2)14/h3-4,6-7,12-13H,5,8-9H2,1-2H3/b4-3+,7-6+/t12-,13+/m0/s1
PTNNGEBMCNMENY-JIVMHGEESA-N
Informazioni sul gene
human ... FASN(2194)
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Descrizione generale
Applicazioni
- as a fatty acid synthase inhibitor to study its effects on aldosterone-induced trained immunity
- as a blocker of fatty acid synthase to study its effects on the replication of severe acute respiratory syndrome coronavirus 2 (SARS-CoV2)
- as a supplement in yeast extract–peptone–dextrose/glycerol (YPD/G) agar plates for the isolation of cerulenin-resistant yeast strains
Avvertenze
Warning
Indicazioni di pericolo
Classi di pericolo
Acute Tox. 4 Oral
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
dust mask type N95 (US), Eyeshields, Gloves
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Articoli
The amount of cholesterol that is synthesized in the liver is tightly regulated by dietary cholesterol levels. LDL receptors regulate the cellular transport of lipid rich low density lipoprotein (LDL) particles.
Information on fatty acid synthesis and metabolism in cancer cells. Learn how proliferatively active cells require fatty acids for functions such as membrane generation, protein modification, and bioenergetic requirements. These fatty acids are derived either from dietary sources or are synthesized by the cell.
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.