40796
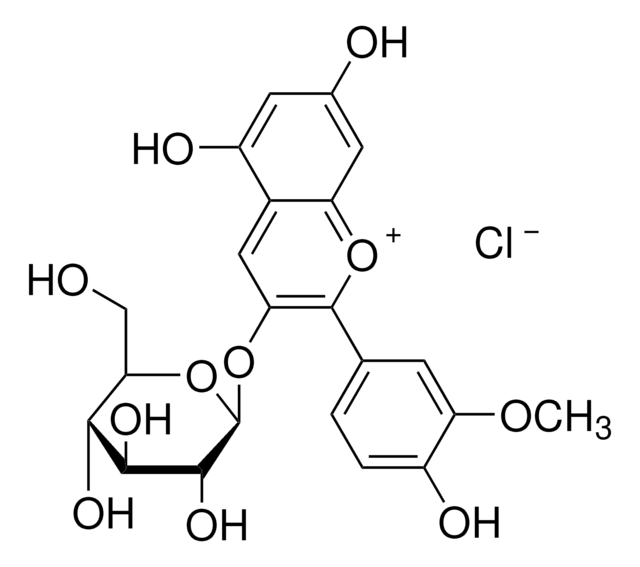
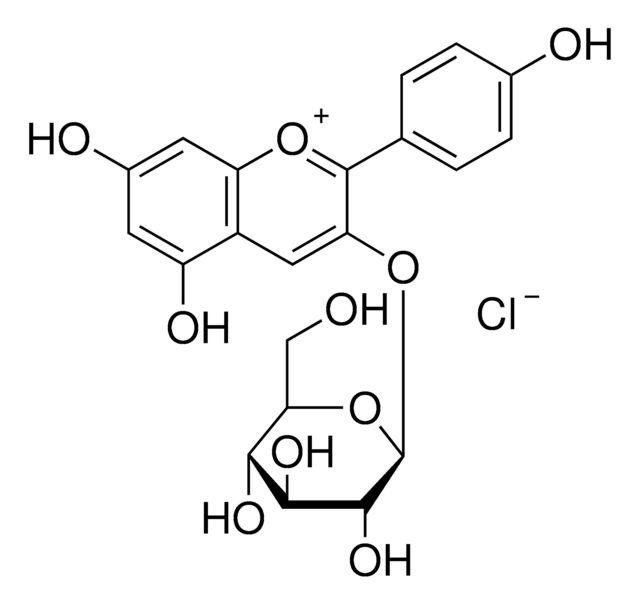
Peonidin 3-O-glucoside chloride
≥95% (HPLC)
Sinonimo/i:
3-(Glucosyloxy)-4′,5,7-trihydroxy-3′-methoxyflavylium chloride, Glucopeonidin chloride
About This Item
Prodotti consigliati
Origine biologica
synthetic
Saggio
≥95% (HPLC)
Stato
powder
applicazioni
metabolomics
vitamins, nutraceuticals, and natural products
Temperatura di conservazione
−20°C
Stringa SMILE
[Cl-].COc1cc(ccc1O)-c2[o+]c3cc(O)cc(O)c3cc2O[C@@H]4O[C@H](CO)[C@@H](O)[C@H](O)[C@H]4O
InChI
1S/C22H22O11.ClH/c1-30-15-4-9(2-3-12(15)25)21-16(7-11-13(26)5-10(24)6-14(11)31-21)32-22-20(29)19(28)18(27)17(8-23)33-22;/h2-7,17-20,22-23,27-29H,8H2,1H3,(H2-,24,25,26);1H/t17-,18-,19+,20-,22-;/m1./s1
VDTNZDSOEFSAIZ-VXZFYHBOSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Descrizione generale
Applicazioni
Azioni biochim/fisiol
Confezionamento
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Protocolli
-glucoside chloride; Malvidin 3-glucoside; Delphinidin 3-(6-acetylglucoside); Cyanidin 3-(6-acetylglucoside); Petunidin 3-(6-acetylglucoside); Peonidin 3-(6-acetylglucoside); Malvidin 3-(6-acetylglucoside); Malvidin 3-(6-caffeoylglucoside); Petunidin 3-(6-cumarylglucoside); Peonidin 3-(6-cumarylglucoside); Malvidin 3-(6-cumarylglucoside)
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.