V0100000
Verapamil hydrochloride
European Pharmacopoeia (EP) Reference Standard
Sinonimo/i:
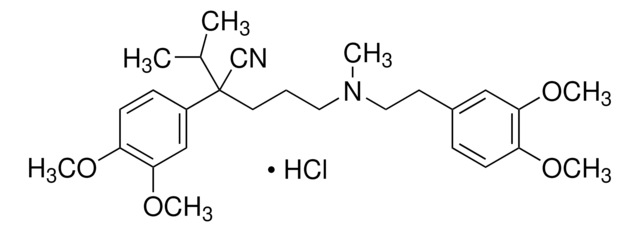
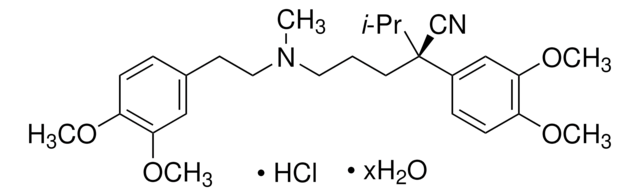
(±)-Verapamil hydrochloride, 5-[N-(3,4-Dimethoxyphenylethyl)methylamino]-2-(3,4-dimethoxyphenyl)-2-isopropylvaleronitrile hydrochloride
About This Item
Prodotti consigliati
Grado
pharmaceutical primary standard
agenzia
EP Reference Standard
Famiglia di API
verapamil
Produttore/marchio commerciale
EDQM
Punto di fusione
142 °C (dec.) (lit.)
applicazioni
pharmaceutical (small molecule)
Formato
neat
Temperatura di conservazione
2-8°C
Stringa SMILE
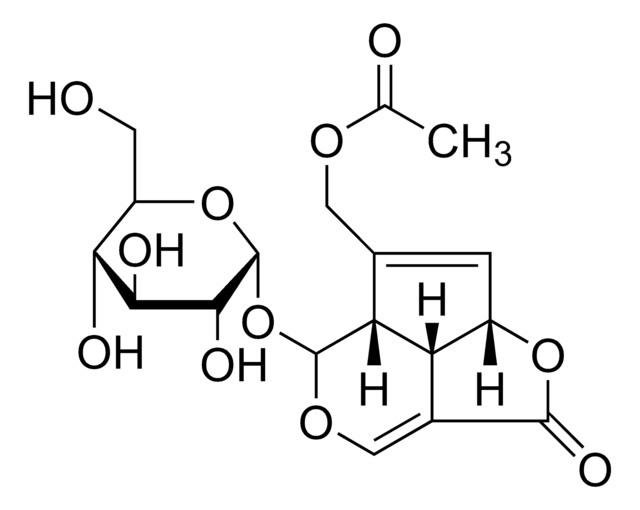
Cl.COc1ccc(CCN(C)CCCC(C#N)(C(C)C)c2ccc(OC)c(OC)c2)cc1OC
InChI
1S/C27H38N2O4.ClH/c1-20(2)27(19-28,22-10-12-24(31-5)26(18-22)33-7)14-8-15-29(3)16-13-21-9-11-23(30-4)25(17-21)32-6;/h9-12,17-18,20H,8,13-16H2,1-7H3;1H
DOQPXTMNIUCOSY-UHFFFAOYSA-N
Informazioni sul gene
human ... CACNA1C(775) , CACNA1D(776) , CACNA1F(778) , CACNA1S(779)
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Descrizione generale
Applicazioni
Azioni biochim/fisiol
Confezionamento
Altre note
Avvertenze
Danger
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Acute Tox. 3 Oral - Aquatic Chronic 2
Codice della classe di stoccaggio
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Scegli una delle versioni più recenti:
Certificati d'analisi (COA)
It looks like we've run into a problem, but you can still download Certificates of Analysis from our Documenti section.
Se ti serve aiuto, non esitare a contattarci Servizio Clienti
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Protocolli
LC/MS/MS Analysis of Interacting Cardiac Drugs Digoxin, Quinidine, Amiodarone and Verapamil on Titan™ C18
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.