F20408
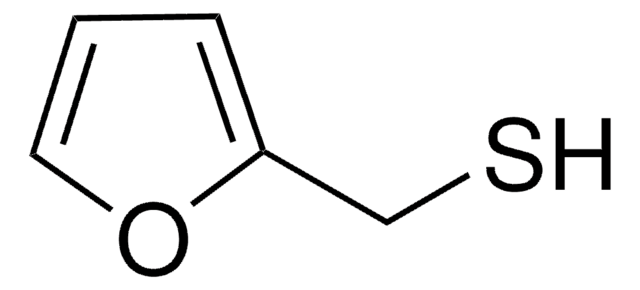
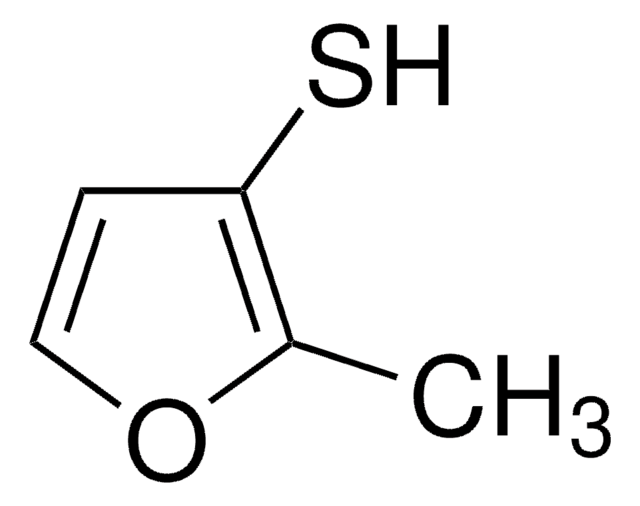
2-Furanmethanethiol
98%
Sinonimo/i:
2-Furfurylthiol, 2-Furylmethanethiol, Furfuryl mercaptan
About This Item
Prodotti consigliati
Livello qualitativo
Saggio
98%
Stato
liquid
Indice di rifrazione
n20/D 1.531 (lit.)
P. ebollizione
155 °C (lit.)
Densità
1.132 g/mL at 25 °C (lit.)
Stringa SMILE
SCc1ccco1
InChI
1S/C5H6OS/c7-4-5-2-1-3-6-5/h1-3,7H,4H2
ZFFTZDQKIXPDAF-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Flam. Liq. 3
Codice della classe di stoccaggio
3 - Flammable liquids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
113.0 °F - closed cup
Punto d’infiammabilità (°C)
45 °C - closed cup
Dispositivi di protezione individuale
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Global Trade Item Number
| SKU | GTIN |
|---|---|
| F20408-25G | 4061833612675 |
| F20408-100G |
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.