D223204
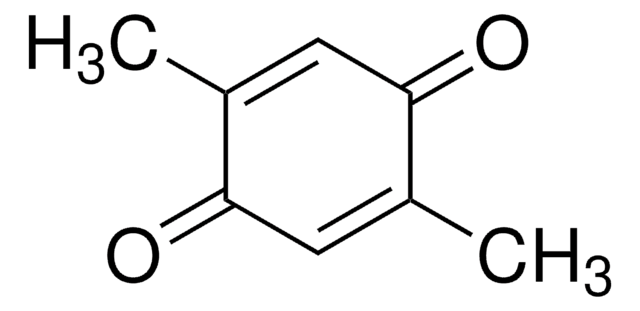
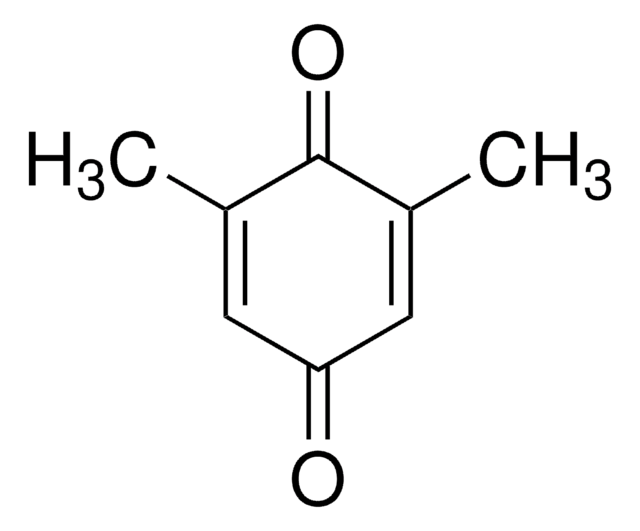
Duroquinone
97%
Sinonimo/i:
2,3,5,6-Tetramethyl-1,4-benzoquinone, Tetramethyl-p-benzoquinone
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula empirica (notazione di Hill):
C10H12O2
Numero CAS:
Peso molecolare:
164.20
Beilstein:
1909128
Numero CE:
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Saggio
97%
Stato
powder
Punto di fusione
110-112 °C (lit.)
Stringa SMILE
CC1=C(C)C(=O)C(C)=C(C)C1=O
InChI
1S/C10H12O2/c1-5-6(2)10(12)8(4)7(3)9(5)11/h1-4H3
WAMKWBHYPYBEJY-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Categorie correlate
Descrizione generale
Duroquinone is an organic oxidant that belongs to the class of 1,4-benzoquinones, is used in redox flow Li–O2 batteries.
Applicazioni
- Role of Duroquinone in Photosynthetic Research: A study on purple bacterial photosynthetic reaction centers highlighted Duroquinone′s role when incorporated into the QA binding site, impacting the isotope edited FTIR difference spectra, crucial for understanding energy conversion processes in photosynthesis (Zhao et al., 2013).
- Duroquinone as a Molecular Ion Source: Research demonstrated the generation of molecular negative ions by Duroquinone, showcasing its potential in mass spectrometry applications for studying molecular ionization and longevity (Khvostenko et al., 2012).
- Duroquinone in Biochemical Sensors: Investigation into the optimization of gold electrode surfaces with photosystem II monolayers revealed Duroquinone′s critical role in enhancing sensor responses, applicable in biochemical sensor technology (Maly et al., 2005).
- Cytotoxicity of Duroquinone Congeners: A comparative study of 14 p-benzoquinone congeners, including Duroquinone, used quantitative structure-toxicity relationships to evaluate cytotoxicity in rat hepatocytes and PC12 cells, relevant for safety assessments in chemical manufacturing (Siraki et al., 2004).
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
dust mask type N95 (US), Eyeshields, Gloves
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Marilyn P Merker et al.
American journal of physiology. Lung cellular and molecular physiology, 290(3), L607-L619 (2005-10-26)
The objective of this study was to examine the impact of chronic hyperoxic exposure (95% O2 for 48 h) on intact bovine pulmonary arterial endothelial cell redox metabolism of 2,3,5,6-tetramethyl-1,4-benzoquinone (duroquinone, DQ). DQ or durohydroquinone (DQH2) was added to normoxic
Bingsheng Zhou et al.
Aquatic toxicology (Amsterdam, Netherlands), 77(2), 136-142 (2006-01-18)
Toxicity of many waterborne organic contaminants to aquatic organisms is mediated through oxidative damages resulting from the production of reactive oxygen species (ROS). Using duroquinone as a model ROS inducer, we carried out in vitro and in vivo experiments to
Said H Audi et al.
American journal of physiology. Lung cellular and molecular physiology, 289(5), L788-L797 (2005-07-05)
NAD(P)H:quinone oxidoreductase 1 (NQO1) plays a dominant role in the reduction of the quinone compound 2,3,5,6-tetramethyl-1,4-benzoquinone (duroquinone, DQ) to durohydroquinone (DQH2) on passage through the rat lung. Exposure of adult rats to 85% O2 for > or =7 days stimulates
Xiao-Hui Duan et al.
The Journal of chemical physics, 120(21), 10025-10032 (2004-07-23)
Photoinduced electron transfer of the model system composed of vitamin E and duroquinone has been investigated using time-dependent density functional theory. Calculations for the excited states tell that the photoexcitation of the model system can directly yield the charge transfer
J Maly et al.
Analytical and bioanalytical chemistry, 381(8), 1558-1567 (2005-04-12)
Mass transport of the bulk of the analyte to the electrode and through the bioactive layer can be significantly improved by use of the nanoelectrode array and defined arrangement of protein film. This phenomenon has been studied by (i) atomic-force
Global Trade Item Number
| SKU | GTIN |
|---|---|
| D223204-25G | |
| D223204-5G | 4061831826548 |
| D223204-1G | 4061825597713 |
| D223204-500G |
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.