C109800
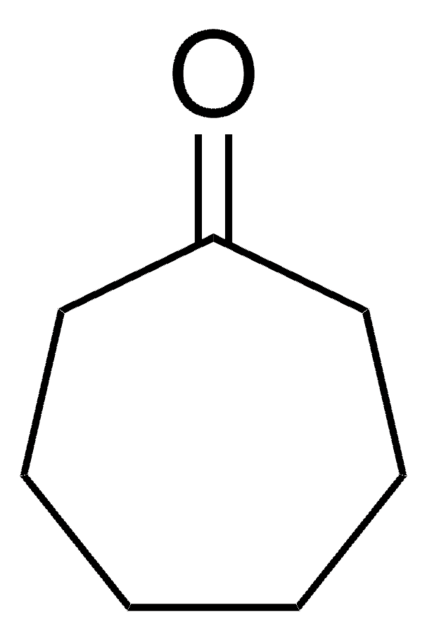
Cyclooctanone
98%
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula condensata:
C8H14(=O)
Numero CAS:
Peso molecolare:
126.20
Beilstein:
1280738
Numero CE:
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Livello qualitativo
Saggio
98%
Stato
crystals
P. ebollizione
195-197 °C (lit.)
Punto di fusione
32-41 °C (lit.)
Densità
0.958 g/mL at 25 °C (lit.)
Stringa SMILE
O=C1CCCCCCC1
InChI
1S/C8H14O/c9-8-6-4-2-1-3-5-7-8/h1-7H2
IIRFCWANHMSDCG-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Avvertenze
Danger
Indicazioni di pericolo
Classi di pericolo
Eye Dam. 1 - Skin Corr. 1B
Codice della classe di stoccaggio
8A - Combustible corrosive hazardous materials
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
165.2 °F
Punto d’infiammabilità (°C)
74 °C
Dispositivi di protezione individuale
Eyeshields, Gloves, type N95 (US)
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Vishwakarma Singh et al.
The Journal of organic chemistry, 70(3), 973-981 (2005-01-29)
A new and efficient synthesis of a variety of highly embellished bicyclooctenones having an endo-vinyl moiety and their sigmatropic shifts in ground and excited states leading to a stereoselective route to substituted cis-decalins and diquinane frameworks have been described. Functionalized
M E Krafft et al.
The Journal of organic chemistry, 66(22), 7443-7448 (2001-10-30)
The total synthesis of asteriscanolide (1) has been achieved by taking advantage on an intermolecular Pauson-Khand cycloaddition and a ring-closing metathesis as key bond-forming transformations. The approach incorporates the cyclooctane stereogenic center prior to ring formation. Interestingly, the ring-closing metathesis
K Yamada et al.
Chemical & pharmaceutical bulletin, 45(12), 1898-1905 (1998-01-20)
Construction of the AB-ring system of the taxane framework via an A-ring annulation strategy was demonstrated by base-mediated intramolecular aldol reaction of (Z)-2,2-dimethyl-3-(1-methyl-2-oxopropylidene)cyclooctanone, affording the title compound, 1-hydroxy-8,11,11-trimethylbicyclo[5.3.1]undec-7-en-9-one. A cyclization precursor, the tetra-substituted (Z)-alkene, was prepared from the corresponding cyclooctanone
F E Harvey et al.
Brain research bulletin, 13(4), 541-547 (1984-10-01)
Female mice were reared in observation incubators from day 1 of life for three weeks. During that time they were continuously exposed to the odors of either cyclooctanone, adult male mouse urine or distilled water. The growth rate was temporarily
K Yamada et al.
Chemical & pharmaceutical bulletin, 45(12), 2113-2115 (1998-01-20)
Stereoselective syntheses of omega-(alpha-bromoketo) octanals and nonanal with oxygenated functions and formation of the corresponding eight-membered carbocyclic aldols by subsequent samarium(II)-mediated cyclization are demonstrated. Cyclooctenones deoxygenated at the C2 or C10 position in the taxane framework are prepared by dehydration
Global Trade Item Number
| SKU | GTIN |
|---|---|
| C109800-25G | 4061833460696 |
| C109800-100G | 4061833460689 |
| C109800-5KG |
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.