576638
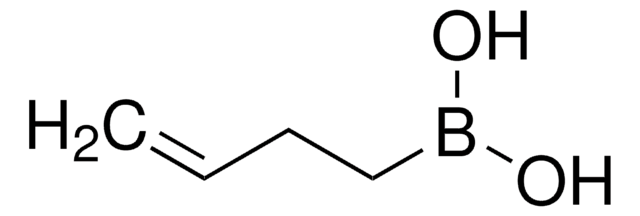
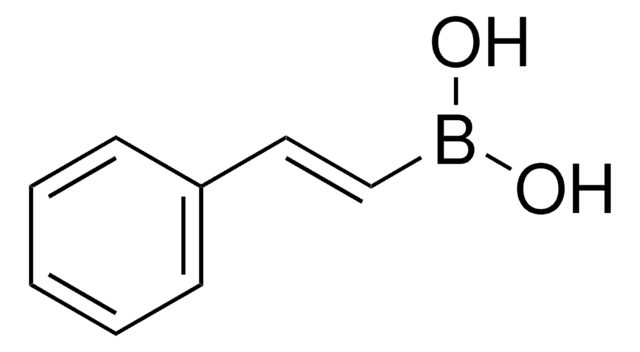
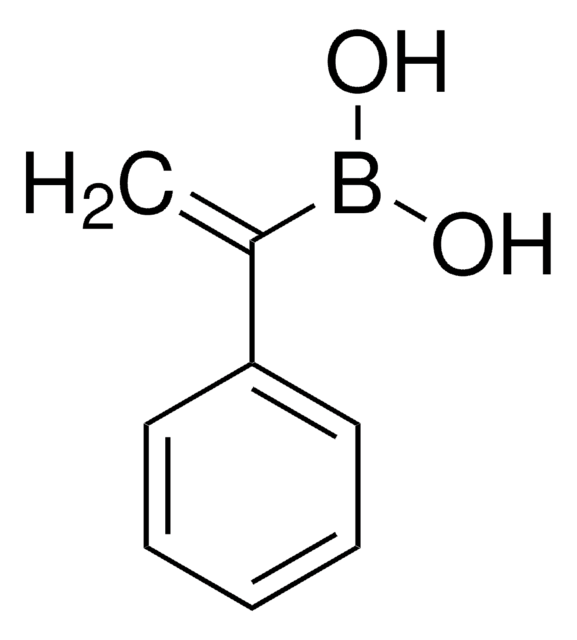
trans-1-Propen-1-ylboronic acid
≥95.0%
Sinonimo/i:
(E)-1-Propen-1-ylboronic acid, (E)-Prop-1-enylboronic acid, trans-1-Propeneboronic acid, trans-1-Propenylboronic acid, trans-Propenylboronic acid
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula condensata:
CH3CH=CHB(OH)2
Numero CAS:
Peso molecolare:
85.90
Numero MDL:
Codice UNSPSC:
12352103
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Saggio
≥95.0%
Impurezze
~10 wt. % cis-isomer
Punto di fusione
123-127 °C (lit.)
Temperatura di conservazione
2-8°C
Stringa SMILE
[H]\C(C)=C(\[H])B(O)O
InChI
1S/C3H7BO2/c1-2-3-4(5)6/h2-3,5-6H,1H3/b3-2+
CBMCZKMIOZYAHS-NSCUHMNNSA-N
Applicazioni
Reactant for:
Reactant for preparation of:
- Palladium-phosphine-catalyzed Suzuki-Miyaura coupling reactions
- Cu(II)-mediated Ullmann-type coupling
Reactant for preparation of:
- Alkynylphenoxyacetic acids as DP2 receptor antagonists for treatment of allergic inflammatory diseases
- Tetrahydrobenzothiophenes as conformationally restricted enol-mimic inhibitors of type II dehydroquinase via Paal-Knorr synthesis involving Suzuki coupling
- Highly substituted benzannulated cyclooctanol derivatives by samarium diiodide-mediated cyclization
- Stereospecific dienes via nickel-catalyzed three-component reductive coupling with alkynes and enones
Reactant for
Reactant for preparation of
- Palladium-phosphine-catalyzed Suzuki-Miyaura coupling reactions
- Cu(II)-mediated Ullmann-type coupling
- Palladium-catalyzed Sonogashira cross-coupling
Reactant for preparation of
- Alkynylphenoxyacetic acids as DP2 receptor antagonists for treatment of allergic inflammatory diseases
- Tetrahydrobenzothiophenes as conformationally restricted enol-mimic inhibitors of type II dehydroquinase via Paal-Knorr synthesis involving Suzuki coupling
- Highly substituted benzannulated cyclooctanol derivatives by samarium diiodide-mediated cyclization
- Stereospecific dienes via nickel-catalyzed three-component reductive coupling with alkynes and enones
Altre note
Contains varying amounts of anhydride
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
Eyeshields, Gloves, type N95 (US)
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Tetrahydrobenzothiophene derivatives: conformationally restricted inhibitors of type II dehydroquinase.
Sonia Paz et al.
ChemMedChem, 6(2), 266-272 (2011-01-29)
The preparation of substituted pyrazoles from β,β-dibromo-enones by a tandem condensation/Suzuki-Miyaura cross-coupling process
Beltran-Rodil, S.; et al.
Synlett, 4, 602-606 (2010)
Highly substituted benzannulated cyclooctanol derivatives by samarium diiodide-induced cyclizations.
Jakub Saadi et al.
Beilstein journal of organic chemistry, 6, 1229-1245 (2011-02-02)
A series of γ-oxo esters suitably substituted with various styrene subunits was subjected to samarium diiodide-induced 8-endo-trig cyclizations. Efficacy, regioselectivity and stereoselectivity of these reactions via samarium ketyls strongly depend on the substitution pattern of the attacked alkene moiety. The
A new strategy for the synthesis of substituted dihydropyrones and tetrahydropyrones via palladium-catalyzed coupling of thioesters
Fuwa, H.; et al.
Tetrahedron, 67, 4995-5010 (2011)
Ming-Bo Zhou et al.
The Journal of organic chemistry, 75(16), 5635-5642 (2010-08-14)
Palladium-catalyzed cross-coupling reaction of terminal alkynes with arylboronic acids has been described. In the presence of Pd(OAc)(2) and Ag(2)O, a variety of terminal alkynes, including electron-poor terminal alkynes, smoothly underwent the reaction with numerous boronic acids to afford the corresponding
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.