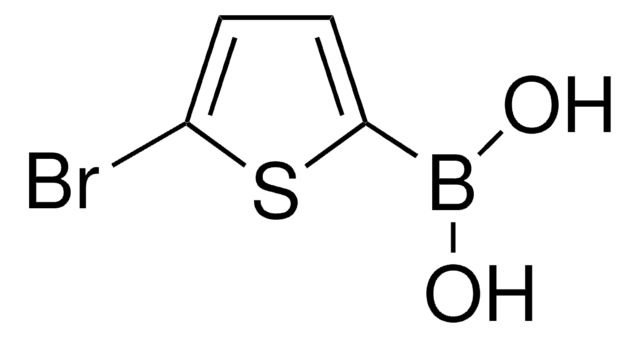
436836
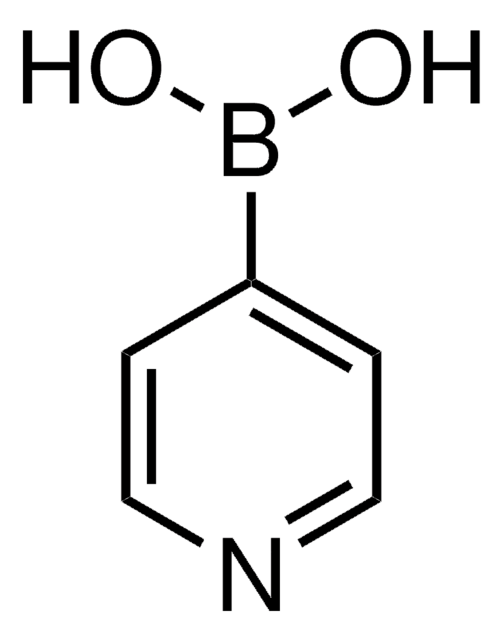
2-Thienylboronic acid
≥95.0%
Sinonimo/i:
2-Thienylboric acid, 2-Thienylboronic acid, Thien-5-ylboronic acid, Thiophene-2-boronic acid
About This Item
Prodotti consigliati
Saggio
≥95.0%
Stato
solid
Punto di fusione
138-140 °C (lit.)
Temperatura di conservazione
2-8°C
Stringa SMILE
OB(O)c1cccs1
InChI
1S/C4H5BO2S/c6-5(7)4-2-1-3-8-4/h1-3,6-7H
ARYHTUPFQTUBBG-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Applicazioni
- Palladium-catalyzed Suzuki-Miyaura cross-couplings
- Alkylation, boration, coupling reaction, Suzuki coupling, and halogenation of fluorenyl bromide
- Chain-growth catalyst transfer polycondensation of conjugated alternating copolymer
- Ferric perchlorate-promoted reaction of fullerene to give fullerenyl boronic esters
- Ligand-free Suzuki, Sonogashira, and Heck cross-coupling reactions
- Copper-catalyzed nitration reactions
- Geometry relaxation-induced Large Stokes shift in red-emitting borondipyrromethenes (BODIPY) and applications in fluorescent thiol probes
Reagent used in Preparation of
- Photophysical properties of oxygen-containing polycyclic aromatic triptycenes
- Donor unit for donor-acceptor-type polymers via N-alkylation, Suzuki coupling, and bromination
- Aminopyridine-based inhibitors of mitotic kinase Nek2 with potential antipoliferative effects in cancer tumors
Altre note
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Organi bersaglio
Respiratory system
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
dust mask type N95 (US), Eyeshields, Gloves
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.
![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)