162698
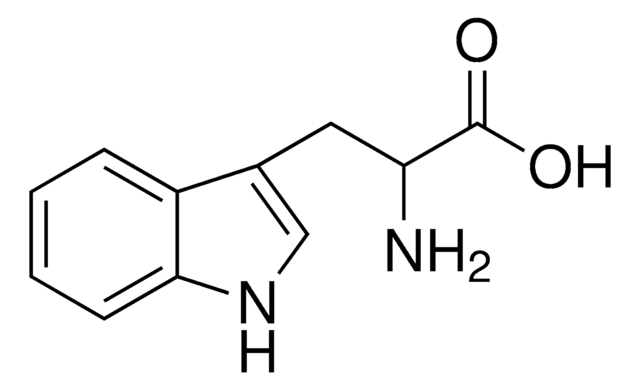
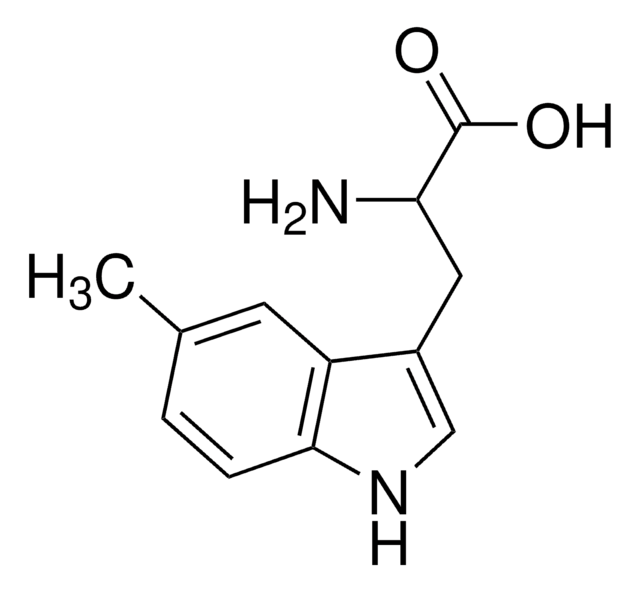
DL-Tryptophan
≥99% (HPLC), for peptide synthesis
Sinonimo/i:
(±)-α-Amino-3-indolepropionic acid, (±)-2-Amino-3-(3-indolyl)propionic acid, DL-3β-Indolylalanine
About This Item
Prodotti consigliati
product name
DL-Tryptophan, ≥99% (HPLC)
Livello qualitativo
Saggio
≥99% (HPLC)
Forma fisica
powder
Impiego in reazioni chimiche
reaction type: solution phase peptide synthesis
Punto di fusione
289-290 °C (dec.) (lit.)
applicazioni
peptide synthesis
Stringa SMILE
NC(Cc1c[nH]c2ccccc12)C(O)=O
InChI
1S/C11H12N2O2/c12-9(11(14)15)5-7-6-13-10-4-2-1-3-8(7)10/h1-4,6,9,13H,5,12H2,(H,14,15)
QIVBCDIJIAJPQS-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Descrizione generale
Applicazioni
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 1
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
Eyeshields, Gloves, type N95 (US)
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.