推荐产品
品質等級
化驗
≥98% (HPLC)
形狀
powder
顏色
white to beige
溶解度
DMSO: 2 mg/mL, clear
儲存溫度
2-8°C
SMILES 字串
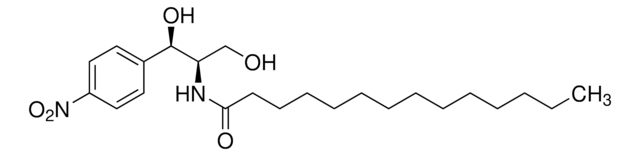
N([C@@H](CO)C[C@H](O)c1ccccc1)C(=O)CCCCCCCCCCC
InChI
1S/C22H37NO3/c1-2-3-4-5-6-7-8-9-13-16-22(26)23-20(18-24)17-21(25)19-14-11-10-12-15-19/h10-12,14-15,20-21,24-25H,2-9,13,16-18H2,1H3,(H,23,26)/t20-,21+/m1/s1
InChI 密鑰
YCAKBKAOFSILDC-RTWAWAEBSA-N
生化/生理作用
(1R,3S)-HPA-12 is a ceramide (Cer) analog that acts as a Cer transporter (CERT) antagonist and selectively inhibits cellular Cer conversion to sphingomyelin (SM), but not to glucosylceramide, by blocking CERT-mediated Cer ER-to-Golgi transport. Common culture dosing range: 1-10 μM.
Ceramide transporter (CERT) antagonist that inhibits Cer-to-sphingomyelin (SM) conversion by blocking CERT-mediated ceramide ER-to-Golgi transport.
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
Masaharu Ueno et al.
Organic letters, 15(11), 2869-2871 (2013-05-25)
In response to Berkeš's report revising the stereochemistry of HPA-12, an important ceramide-trafficking inhibitor that was discovered and synthesized and its stereochemistry determined in 2001, the synthesis and the stereochemistry were reinvestigated. A large-scale synthetic method for HPA-12 based on
Hannah Scheiblich et al.
Journal of neurochemistry, 143(5), 534-550 (2017-09-25)
Inflammation within the CNS is a major component of many neurodegenerative diseases. A characteristic feature is the generation of microglia-derived factors that play an essential role in the immune response. IL-1β is a pro-inflammatory cytokine released by activated microglia, able
S Yasuda et al.
The Journal of biological chemistry, 276(47), 43994-44002 (2001-09-08)
Ceramide produced at the endoplasmic reticulum (ER) is transported to the lumen of the Golgi apparatus for conversion to sphingomyelin (SM). N-(3-Hydroxy-1-hydroxymethyl-3-phenylpropyl)dodecanamide (HPA-12) is a novel analog of ceramide. Metabolic labeling experiments showed that HPA-12 inhibits conversion of ceramide to
Andrej Ďuriš et al.
Organic letters, 13(7), 1642-1645 (2011-03-01)
The practical stereodivergent route to both syn- and anti-diastereomers of 1-substituted 3-aminobutane-1,4-diols based on the crystallization-induced asymmetric transformation (CIAT) approach was completed. This led to the revision of the reported stereochemistry of the first inhibitor of CERT-dependent ceramide trafficking HPA-12
Yasuhiro Hayashi et al.
The Journal of biological chemistry, 293(45), 17505-17522 (2018-09-23)
Sphingolipids, including sphingomyelin (SM) and glucosylceramide (GlcCer), are generated by the addition of a polar head group to ceramide (Cer). Sphingomyelin synthase 1 (SMS1) and glucosylceramide synthase (GCS) are key enzymes that catalyze the conversion of Cer to SM and
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门