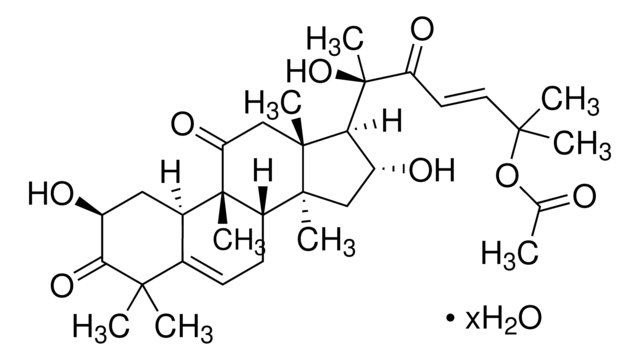
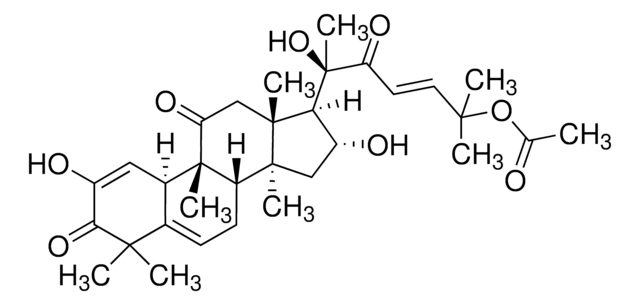
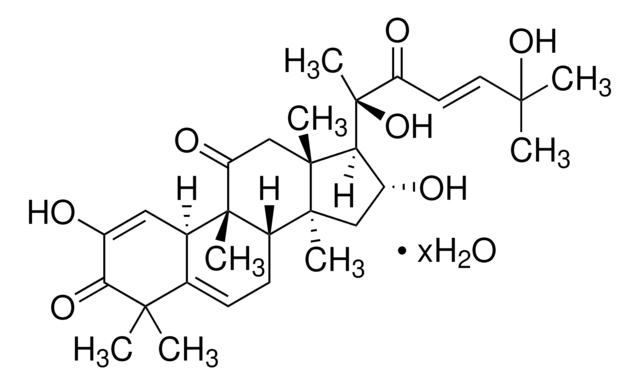
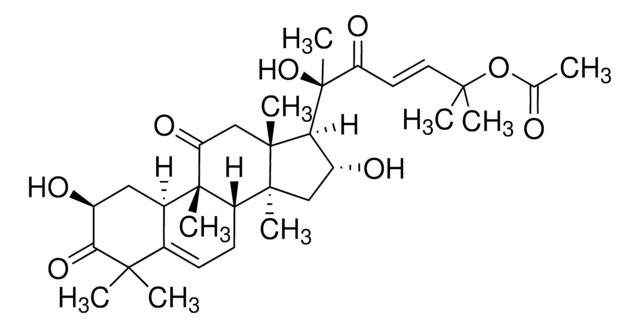
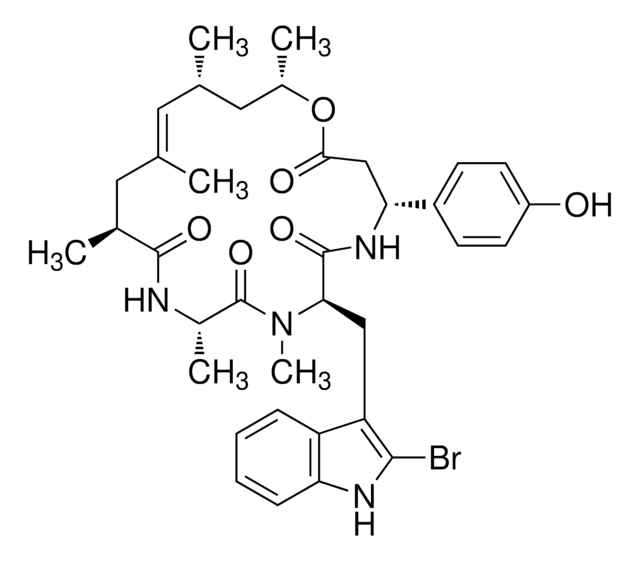
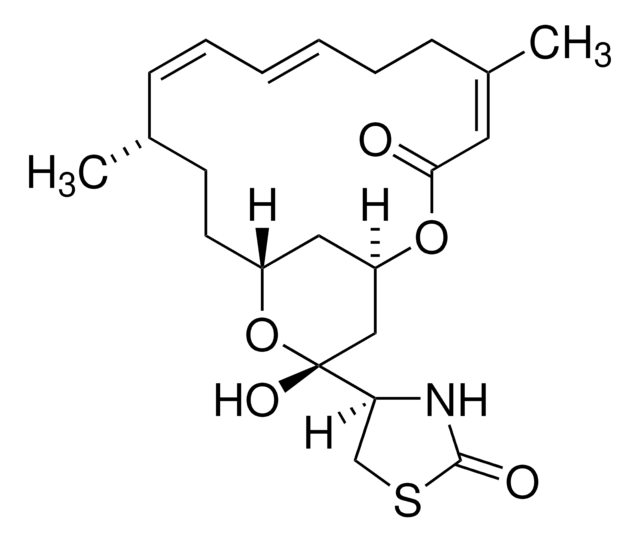
推荐产品
品質等級
化驗
≥95% (HPLC)
形狀
powder
光學活性
[α]/D -60 to -75°, c = 0.7 (CDCl3)
顏色
white to beige
溶解度
DMSO: 15 mg/mL, clear
儲存溫度
−20°C
InChI
1S/C32H44O8/c1-17(33)40-27(2,3)13-12-23(36)32(9,39)25-21(35)15-29(6)22-11-10-18-19(14-20(34)26(38)28(18,4)5)31(22,8)24(37)16-30(25,29)7/h10,12-14,19,21-22,25,34-35,39H,11,15-16H2,1-9H3/b13-12+/t19-,21-,22+,25+,29+,30-,31+,32+/m1/s1
InChI 密鑰
NDYMQXYDSVBNLL-MUYMLXPFSA-N
正在寻找类似产品? 访问 产品对比指南
應用
Cucurbitacin E has been used as a cofilin inhibitor. It is also used as a F-actin stabilizer to prevent membrane-associated periodic skeleton (MPS) loss and protect from axonal fragmentation.
生化/生理作用
Cucurbitacin E is a potent inhibitor of actin depolymerization. Cucurbitacin E is more active than jasplakinolide, and has a different mechanism of action, binding to a different site. Cucurbitacin E binds specifically to filamentous actin (F-actin) forming a covalent bond at residue Cys257, but not to monomeric actin (G-actin), stabilizing F-actin, without affecting actin polymerization or nucleation.
訊號詞
Warning
危險聲明
危險分類
Acute Tox. 4 Oral
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
其他客户在看
Pei-Lin Wu et al.
Chemical & pharmaceutical bulletin, 52(3), 345-349 (2004-03-03)
Three new compounds: begonanline (1). nantoamide (2). and methyl (S)-glycerate (3). as well as forty-four known compounds have been isolated and characterized from the rhizomes of Begonia nantoensis. The structures of these compounds were determined by spectral analyses and/or X-ray
Manali Dimri et al.
Development (Cambridge, England), 144(14), 2595-2605 (2017-07-20)
The intrahepatic biliary network is a highly branched three-dimensional network lined by biliary epithelial cells, but how its branching patterns are precisely established is not clear. We designed a new computer-based algorithm that quantitatively computes the structural differences of the
Yasuyuki Sadzuka et al.
International journal of pharmaceutics, 354(1-2), 63-69 (2007-12-07)
We screened various food components for their ability to inhibit doxorubicin (DOX) permeability in tumor cells in vitro with the aim of finding novel modulators. Capsaicin did not change DOX permeability in the tumor cells, although the capsaicin derivatives gingerol
Tehila Tannin-Spitz et al.
Biochemical and biophysical research communications, 364(1), 181-186 (2007-10-19)
The cucurbitacins are of great interest because of the wide range of biological activities they exhibit in plants and animals. We studied the antioxidant properties of cucurbitacin B + E glucosides (cucurbitacin glucoside combination, CGC) and their direct free-radical scavenging
Yanmin Dong et al.
Carcinogenesis, 31(12), 2097-2104 (2010-08-25)
Cucurbitacin E (CuE, α-elaterin), a tetracyclic triterpenes compound from folk traditional Chinese medicine plants, has been shown to inhibit cancer cell growth, inflammatory response and bilirubin-albumin binding. However, the effects of CuE on tumor angiogenesis and its potential molecular mechanism
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门