About This Item
推荐产品
化驗
≥98%
形狀
powder
溶解度
water: 50 mg/mL, clear, colorless to light yellow
儲存溫度
−20°C
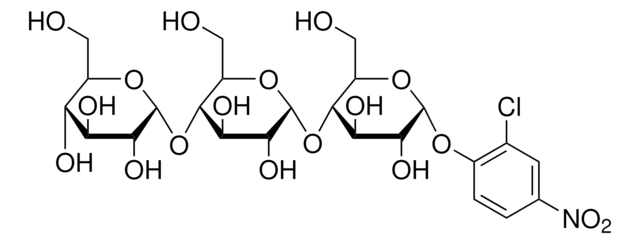
SMILES 字串
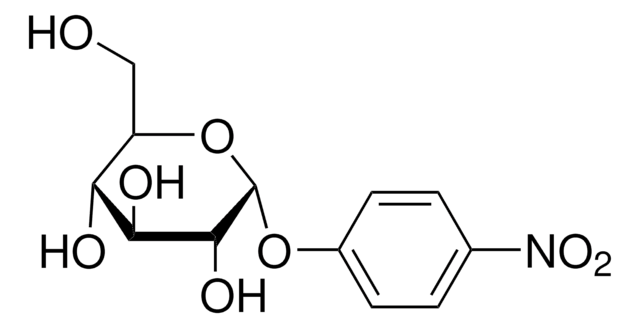
OC[C@H]1O[C@H](O[C@H]2[C@H](O)[C@@H](O)[C@H](O[C@@H]2CO)O[C@H]3[C@H](O)[C@@H](O)[C@H](O[C@@H]3CO)O[C@H]4[C@H](O)[C@@H](O)[C@H](O[C@@H]4CO)O[C@H]5[C@H](O)[C@@H](O)[C@H](O[C@@H]5CO)Oc6ccc(cc6)[N+]([O-])=O)[C@H](O)[C@@H](O)[C@@H]1O
InChI
1S/C36H55NO28/c38-5-12-17(43)18(44)23(49)33(57-12)62-29-14(7-40)59-35(25(51)20(29)46)64-31-16(9-42)61-36(27(53)22(31)48)65-30-15(8-41)60-34(26(52)21(30)47)63-28-13(6-39)58-32(24(50)19(28)45)56-11-3-1-10(2-4-11)37(54)55/h1-4,12-36,38-53H,5-9H2/t12-,13-,14-,15-,16-,17-,18+,19-,20-,21-,22-,23-,24-,25-,26-,27-,28-,29-,30-,31-,32+,33-,34-,35-,36-/m1/s1
InChI 密鑰
YXGBAQKCCMQLGH-MYPSSPKESA-N
正在寻找类似产品? 访问 产品对比指南
基底
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
個人防護裝備
dust mask type N95 (US), Eyeshields, Gloves
其他客户在看
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门